Beijing Kanglewis Biotechnology Co., Ltd. (Stock Code:833575.BJ) successfully listed on the Beijing Stock Exchange on March 15, 2023. Kanglewis is committed to the research, development, and industrialization of innovative vaccines, possessing the richest HPV vaccine portfolio in the world and ranking among the top tier in domestic human papillomavirus (HPV) vaccine development. Frost & Sullivan (Frost &Sullivan, hereinafter referred to as 'Frost & Sullivan'), hereby warmly congratulates the company on its successful listing.
Beijing Kanglewis Biotechnology Co., Ltd. (hereinafter referred to as“Kanglewis”) successfully listed on March 15, 2023, with an issued share number of 8,050,000 shares (after fully exercising the over-allotment option), at an issue price of RMB 42.00 per share.
Investment Highlights
· The company is committed to the research and development of innovative recombinant protein vaccines, possessing a complete vaccine product system, including recombinant trivalentHPV vaccines, female-specific nine-valent HPV vaccines, male-specific nine-valent HPV vaccines, fifteen-valent HPV vaccines, bivalent COVID-19 vaccines, multivalent norovirus vaccines, respiratory syncytial virus vaccines, herpes zoster vaccines, hand-foot-and-mouth disease vaccines, and polio vaccines, etc.;
· The company has currently established multiple key technology platforms, including a structural-based antigen design technology platform, genetic engineering and protein expression technology platform, vaccine engineering technology platform, and recombinant vaccine efficacy evaluation technology platform, and owns three expression systems for Escherichia coli, yeast cells, andCHO cells;
· The company has constructed a complete intellectual property system, owning33 invention patents;
· The company is the first stock in the Beijing Stock Exchange for human vaccines and also the first innovative biopharmaceutical stock to be declared under the fourth set of standards since the opening of the market.
Industry Position
After the outbreak of the COVID-19 pandemic in 2020, the demand for virus sample preservation reagents increased rapidly, making it a major product type among pathogen preservation reagents; The company is the only HPV vaccine R&D enterprise in China that has initiatedclinical trials for male-specific HPV vaccines, in addition to Merck.
Frost & Sullivan has long been paying attention to the global and Chinese biopharmaceutical industries, publishing a large number of research reports, which are widely cited in leadingA-share listed companies' prospectus documents to help customers accelerate growth.
Overview of the Chinese Biopharmaceutical Market
The scale of the Chinese biopharmaceutical market increased from183.6 billion yuan in 2016 to 445.7 billion yuan in 2020, with an annual compound growth rate of 17.1%. Based on academic theory research and years of R&D investment accumulation, the biopharmaceutical market has developed rapidly in recent years, especially showing ideal effects in the prevention and treatment of chronic diseases and gradually becoming a routine preventive measure.
With continuous investment in innovative research and development, the development of innovative biopharmaceutical preparation processes, the follow-up of biosimilars, and with the support of relevant policies, the Chinese biopharmaceutical market will maintain a rapid growth trend. It is expected that by2025 and 2031, the scale of the Chinese biopharmaceutical market will grow to 811.6 billion yuan and 1397.8 billion yuan, with annual compound growth rates of 18.6% and 9.5%, respectively.
Overview of the Chinese Vaccine Market
China isthe second largest vaccine market in the world in 2020. Driven by increased accessibility of innovative vaccines, favorable government policies, vaccine technology innovation, and enhanced vaccination awareness, the Chinese vaccine market increased from RMB 27.1 billion in 2016 to RMB 753 billion in 2020, with a compound annual growth rate of 29.10%, and is expected to reach RMB 383.5 billion in 2031, with a compound annual growth rate of 15.95% from 2020 to 2031.
In recent years, the proportion of vaccine products in the Chinese pharmaceutical market has shown an upward trend, mainly driven bythe outbreak and recurrence of the COVID-19 pandemic, the improvement of China's national economic level and health awareness, etc. In 2020, China's research and development and vaccination progress of novel coronavirus vaccines were rapid, laying a solid foundation for increasing the proportion of vaccines in the entire pharmaceutical market. Driven by potential factors such as continuous new product launches, increased vaccination awareness among residents, and expansion of the national immunization program, the proportion of vaccine products will continue to increase in the future.
Market Scale and Forecast of Chinese Vaccines
From 2016 to 2031 (estimated)
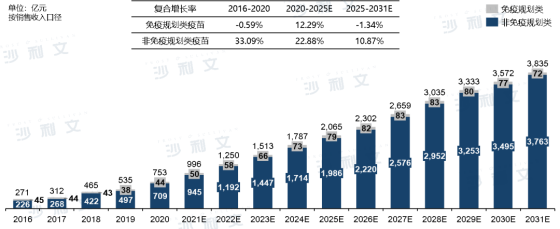
Source: Frost & Sullivan report
ChinaHPV Vaccine Market
Industry Overview
From 2017 to 2020, the market scale of HPV vaccines in China (calculated by batch issuance and winning bid prices) began to grow from RMB 940 million to RMB 1.356 billion, with an annual compound growth rate of 143.4%, mainly benefiting from the double growth in new product approvals and HPV vaccination demand. It is expected that the market scale of HPV vaccines in China will reach RMB 22.97 billion by 2025, with an annual compound growth rate of 11.1%.
Driven by factors such as the successive approval and launch of high-priced local products, China's responseto the WHO's cervical cancer elimination campaign, and the opening of the male vaccination market potential, it is expected that the Chinese HPV vaccine market will enter a period of high-speed growth after 2025, reaching a market scale of RMB 62.54 billion by 2031, with an annual compound growth rate of 18.2% since 2025.
ChinaHPV Vaccine Market Scale and Forecast
From 2016 to 2031 (estimated)
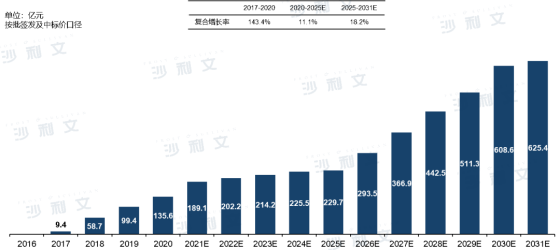
Source: Frost & Sullivan report
Competitive Landscape
By batch issuance caliber, Merckstill accounted for nearly 80% of the HPV market share in 2020. With only four-valent and nine-valent HPV vaccine products in the market, Merck's total four-nine-valent batch issuance volume reached 12,285,838 doses. Wantai Biotech's Xinkening had 46 batches issued, the largest number of batches for all HPV products, with an issuance volume of 2,456,141 doses. GlaxoSmithKline's HPV product line is single, with only 3 batches issued in 2020, totaling only 6.8968 billion doses, about 28% of Wantai Xinkening's issuance volume.
It is expectedthat the distribution pattern of HPV vaccine batches in China in 2021 will change significantly compared to 2020, with the proportion of Merck's four-valent and nine-valent vaccines declining, while the proportion of Wantai Biotech and GlaxoSmithKline's bivalent vaccines reaching 39.2% and 7.8%, respectively, showing significant growth, mainly benefiting from the promotion strategy of low-price vaccines and the implementation of free vaccination programs for eligible women in some cities.
ChinaHPV Vaccine Market Competitive Landscape (by batch issuance volume caliber)
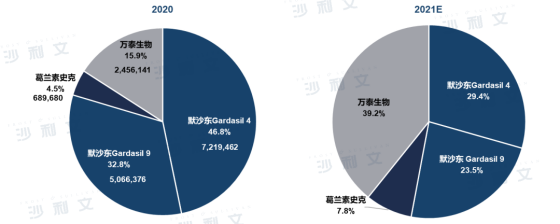
Source: Frost & Sullivan report
Overview of the Chinese Norovirus Vaccine Market
With the advancement of the R&D progress and clinical progress of the Chinese norovirus vaccine pipeline, it is expected that relevant products will be approved and launched as early as2026. The enhanced health protection awareness of residents helps to increase the willingness and rate of norovirus vaccination, and it is expected that from 2026 to 2031, the market scale of Chinese norovirus vaccines can grow from 1.53 billion yuan to 22.93 billion yuan, with an annual compound growth rate of 71.9%.
Overview of the Chinese Norovirus Vaccine Market Scale and Forecast
From 2016 to 2031 (estimated)
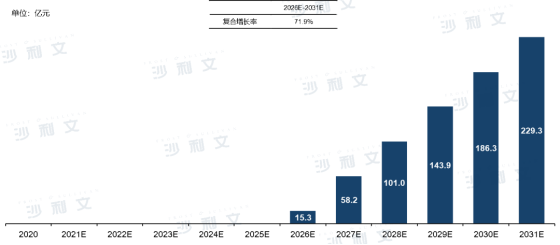
Source: Frost & Sullivan report
Overview of the Chinese Respiratory Syncytial Virus Vaccine Market
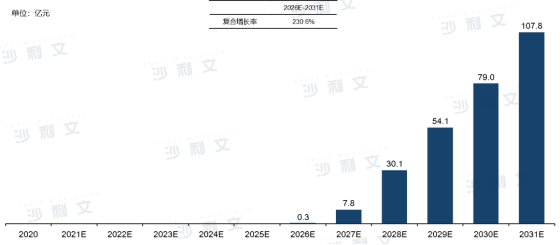
Due to the gap in the respiratory syncytial virus vaccine field, although China currently does not have a relevant clinical pipeline, products with faster clinical progress abroad will have the opportunity to be introduced earlier after launch. In addition, several Chinese vaccine companies have also started to layout the research and development of respiratory syncytial virus vaccines. It is expectedthat from 2026 to 2031, the Chinese respiratory syncytial virus vaccine market can grow from 0.3 billion yuan to 10.78 billion yuan, with an annual compound growth rate of 230.6%.
Overview of the Chinese Respiratory Syncytial Virus Vaccine Market Scale and Forecast
From 2016 to 2031 (estimated)
Source: Frost & Sullivan report

