Ying'en Biotech Co., Ltd. (Stock Code: 9606.HK) successfully listed on the main board of the Hong Kong capital market on April 15, 2025. The company is a key leader in the global antibody conjugate drug (ADC) field, dedicated to developing innovative ADC drugs for patients with cancer and autoimmune diseases. Frost & Sullivan (hereinafter referred to as 'Frost & Sullivan') provides exclusive industry advisory services for the listing of Ying'en Biotech Co., Ltd., and hereby warmly congratulates them on their successful listing.

Yingen Biotech Co., Ltd. (hereinafter referred to as 'Yingen Biotech') successfully listed on April 15, 2025. The company plans to issue 1,507.16 million H shares, of which 90% will be international offerings and 10% will be public offerings. The maximum offering price per share is HK$103.20, raising a net amount of approximately HK$1.555 billion.
During the process of listing in Hong Kong this time, Frost & Sullivan mainly undertook the following tasks: helping the issuer accurately and objectively understand its positioning in the target market, using objective market data to discover, support and highlight the issuer's competitive advantages, assisting the issuer, investment banks and other intermediaries in completing the writing of relevant parts of the prospectus (such as overview, competitive advantages and strategy, industry overview, business and other important chapters), helping the issuer complete communication with the Hong Kong Stock Exchange and investors, assisting investors in quickly understanding the market ecosystem and competitive landscape, and assisting the issuer in completing feedback on various industry-related issues from the Hong Kong Stock Exchange.
According to LiveReport's big data (statistical data as of April 3, 2025), in the first quarter of 2025, as well as during the past 12 and 36 months, Frost & Sullivan provided listing industry advisory services to 11 (73%), 47 (64%), and 147 (68%) Hong Kong-listed IPOs respectively, boasting rich industry experience and communication skills with exchanges and investors.
PART/1
Investment Highlights
-
As a major player in the global ADC field, the company possesses insights that can lead ADC innovation and strong execution capabilities;
-
The company's core product DB-1303/BNT323 is a globally leading HER2 ADC candidate for the treatment of endometrial cancer (EC) with varying HER2 expression levels, and it is also a leading candidate for clinical development in HER2-low breast cancer (BC).
-
The company's core product DB-1311/BNT324 is a B7-H3 ADC candidate drug with a globally leading clinical progress;
-
The company's key product DB-1310 is an HER3 ADC candidate drug in Phase I/IIa clinical trials and is one of the world's leading HER3 ADC candidates in clinical trials;
-
The company's key product, DB-1305/BNT325, is a TROP2 ADC candidate with potential as a frontline pillar therapy;
-
The company's key product, DB-1419, is an innovative B7-H3xPD-L1 bispecific ADC candidate drug that connects a DNA topoisomerase I inhibitor and is currently the only B7-H3xPD-L1 bispecific ADC in clinical development worldwide.
-
The company's key product, DB-2304, is an innovative BDCA2 ADC candidate drug for the treatment of systemic lupus erythematosus (SLE) and cutaneous lupus erythematosus (CLE);
-
The company innovates ADC development with a multi-functional platform, covering therapeutic areas where needs are not yet met;
-
The company has established strategic and value-enhancing partnerships, demonstrating confidence in its platform and pipeline, and supporting its long-term development globally;
-
The company is led by a world-class management team composed of ADC experts and experienced entrepreneurs, with a solid track record.
PART/2
Global ADC Industry Market Overview
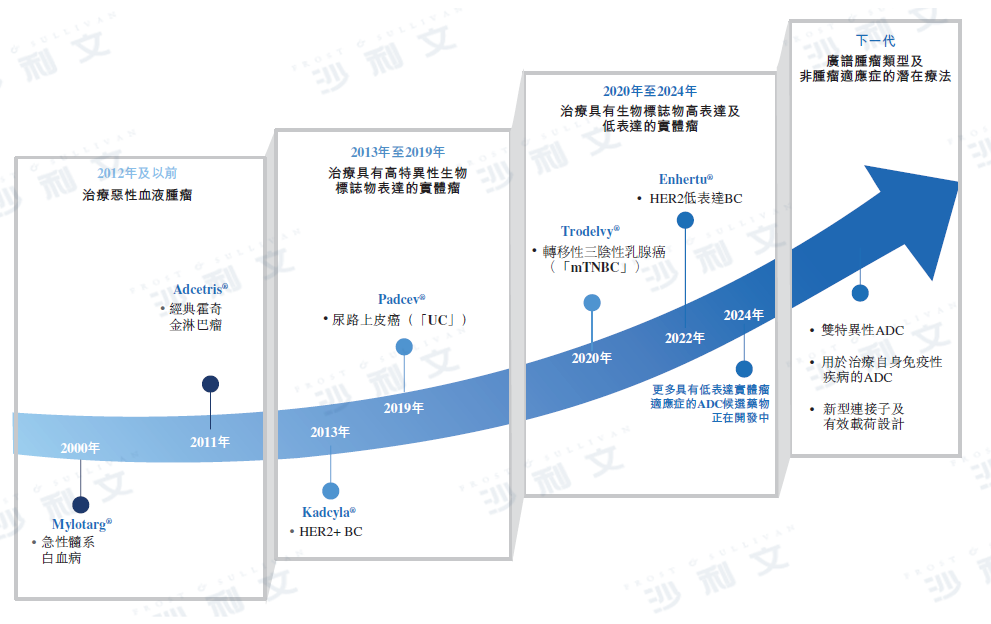
Antibody-drug conjugates (ADCs) have become one of the most promising and rapidly developing treatment modalities, utilizing the targeting and binding capabilities of antibodies to precisely deliver cytotoxic payloads to cancer cells or other diseased cells. The first ADC was approved in 2000, and among the 16 ADCs that have been marketed to date, 11 were approved after 2019, with five of them becoming blockbuster products (i.e., with annual sales exceeding $1 billion).
Despite the tremendous therapeutic potential of ADCs, early ADCs faced various challenges, including intolerable toxicity and poor efficacy, which hindered many ADC development projects from the 1980s to the early 2020s. Mylotarg®(The first ADC) was approved by the US Food and Drug Administration (FDA) in 2000 for the treatment of acute myeloid leukemia. Since then, ADC technology has undergone continuous innovation, achieving substantial improvements in stability, tolerability, and efficacy. Specific examples include the introduction of bystander killing effects through new payloads with better transcellular permeability, the evolution from chimeric antibodies to humanized antibodies, and the release of active agents through cleavable linkers in the tumor microenvironment.
It is expected that the next generation ADCs will utilize novel conjugates and payloads, as well as innovative payload molecules superior to traditional cytotoxic drugs, such as immunomodulatory payloads. The innovative frontiers in ADC development also include exploring novel bispecific and multispecific antibody molecules, as well as potential combination therapies that produce synergistic effects with other treatment modalities. All these advancements will pave the way for the development of ADCs, expanding their application scope to frontline treatment and beyond oncology.
The following table provides details on ADC development, representative products, and their therapeutic potential.
Source: FDA, China Food and Drug Administration, Frost & Sullivan analysis
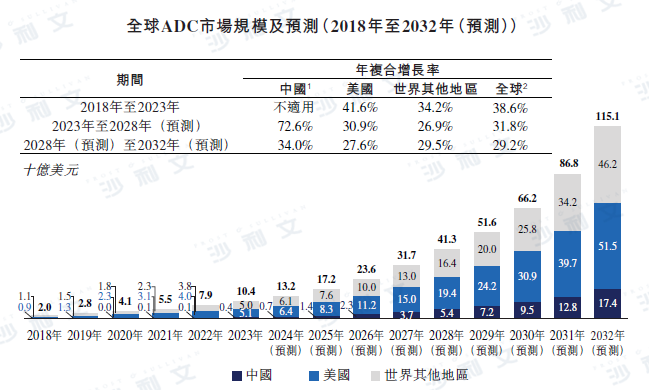
The global ADC market grew rapidly from $2 billion in 2018 to $10.4 billion in 2023, with a compound annual growth rate of 38.6%. It is expected that from 2023 to 2028 and from 2028 to 2032, the market will continue to grow strongly at a compound annual growth rate of 31.8% and 29.2%, respectively, reaching $115.1 billion in 2032. It is expected that the United States and China will remain the largest and fastest-growing ADC markets, with annual compound growth rates of 30.9% and 72.6% from 2023 to 2028. In addition, with the exploration of this form in non-tumor indications, ADCs for autoimmune diseases are expected to further expand the ADC market. The following chart illustrates the growth of the global ADC market by major region.
Data sources: Annual reports of listed healthcare companies, China Food and Drug Administration, Center for Drug Evaluation, National Medical Insurance Drug List, Ministry of Human Resources and Social Security, analysis by Frost & Sullivan
PART/3
ADC Market Drivers
● A patient population with unmet needs and growing demands
In 2023, the global number of cancer cases reached 2080 million, and it is expected to increase to 2550 million by 2032. The number of cases for the main tumor types covered by approved ADCs is currently on the rise, including several subtypes of breast cancer (BC) and non-small cell lung cancer (NSCLC). The global incidence of other cancers such as endometrial cancer (EC), ovarian cancer (OC), small cell lung cancer (SCLC), and castration-resistant prostate cancer (CRPC) is also increasing. Due to the lack of effective treatment methods, as well as the emergence of resistance and recurrence, the five-year survival rate for cancer remains low, highlighting the need for new therapies that can improve cancer prognosis and efficacy. It is noteworthy that ADCs, due to their antibody specificity and the potent cytotoxic ability of cytotoxic drugs, have become a promising upgrade to chemotherapy in cancer therapy, bringing significant market opportunities.
● Expand application scope through technological progress
A large amount of investment has been made in cancer research and drug development with the aim of further elucidating disease biology and discovering cancer-targeted therapies that can improve patient treatment outcomes. In particular, the ongoing research and development of new effective payloads for ADCs may lead to new designs, thereby enhancing the therapeutic effect of this drug form and reducing the toxicity that limits the use of certain marketed ADCs. To date, more than 100 ADC candidate drugs targeting new indications not covered by approved targeted ADCs are in clinical development globally. These efforts will drive ADCs to become the backbone of cancer treatment and expand their application to other therapeutic areas.
● Dynamic cooperation among market participants
The cooperation and licensing transactions in the ADC industry have surged, with large multinational corporations increasing their investment in this field. At the same time, small biotech companies have also made significant contributions to the research and development of ADC candidate drugs. Biotech companies typically use their innovation capabilities and expertise for preliminary exploratory work and concept validation studies, while collaborating with multinational corporations to obtain substantial technical, financial, and regulatory support, thereby accelerating the further development and commercialization of promising ADC candidate drugs. Moreover, the licensing of components used for innovative drug development has become a common practice, especially for complex therapies such as ADCs. The strengthening of cooperation among biotech companies to advance the research and development and commercialization of ADCs has also greatly promoted the growth of the ADC market. For example, in June 2024, MabCare Therapeutics signed a licensing agreement with Day One Biopharma for a PTK7 ADC candidate drug. In December 2024, Innovent Biologics signed a cooperation and licensing agreement with Avenzo for the research and development and commercialization of an EGFR/HER3 bispecific ADC candidate drug. Overall, the collaborative cooperation model is increasingly helping to bring new candidate drugs to market.
PART/4
Global HER2 ADC Market Size
HER2 is a cell surface receptor protein within the HER family, playing a key role in regulating cell growth, division, and survival. After activation through ligand binding or overexpression, HER2 dimerizes with other members of the HER family, leading to the activation of downstream signaling cascades such as the PI3K/AKT and MAPK/ERK pathways. These pathways promote cell proliferation, inhibit apoptosis, and enhance cell migration and invasion. HER2 is expressed at low levels in normal tissues but is abnormally activated in tumor cells through overexpression, promoting tumor cell growth and survival, thereby driving the development of various cancer types. HER2 has become a mature cancer drug target, and different approaches to HER2-targeted therapy have been successful. One of the most successful strategies is HER2 ADCs.
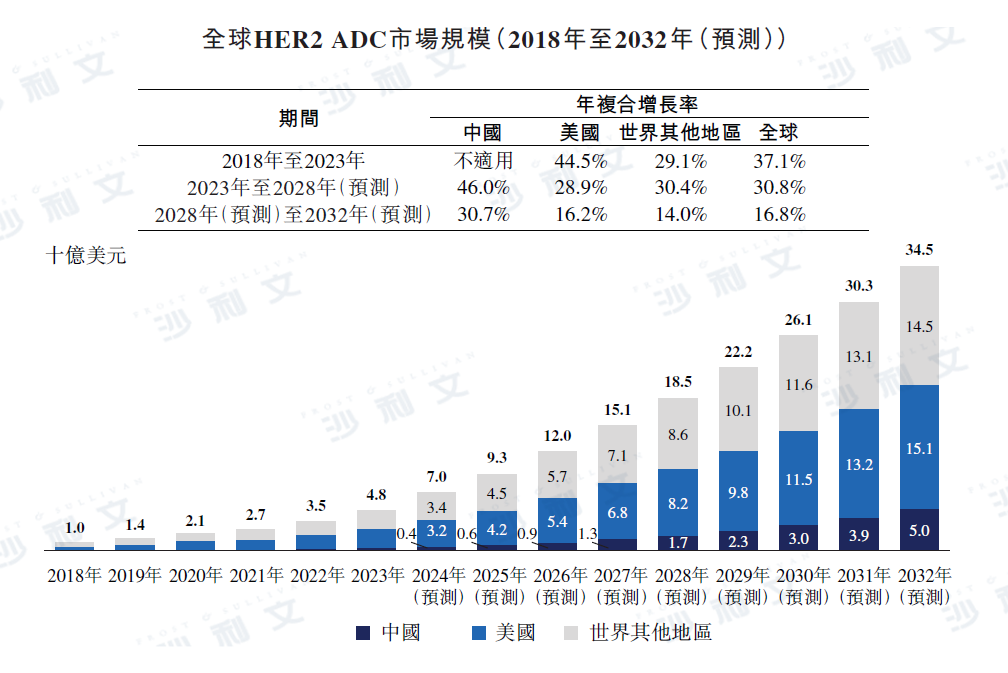
The world's first HER2-targeted ADC, Kadcyla®, was approved in 2013. As shown in the figure below, the global HER2 ADC market increased to $48 billion in 2023, with an annual compound growth rate of 37.1% since 2018. The global HER2 ADC market is expected to grow at an annual compound rate of 30.8% from 2023 to 2028 and from 2028 to 2032, reaching $345 billion by 2032. The table below shows the growth of the global HER2 ADC market by major region.
Data source: Analysis by Frost & Sullivan
PART/5
HER2 ADC Competitive Landscape
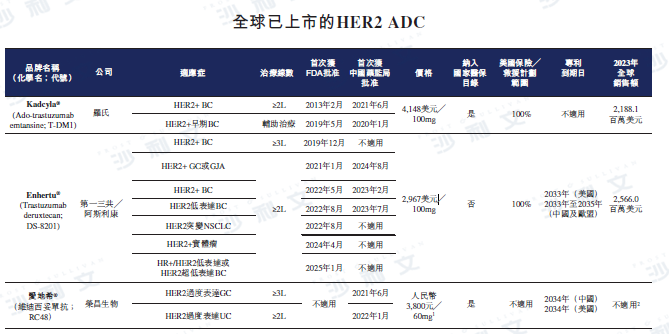
As of the latest practicable date, there are two HER2 ADCs (i.e., Enhertu®And Kadcyla®) has been approved, while in China there is another HER2 ADC (i.e., Aidiex®) have been approved. As of the same day, three HER2 ADCs (including Enhertu®) are in Phase III clinical development or later stages of global multi-region clinical trials (MRCT). The table below describes the competitive landscape of marketed HER2 ADCs and HER2 ADCs in Phase II clinical development or later stages.
Data sources: FDA, China Food and Drug Administration, Drug.com, annual reports, Frost & Sullivan analysis
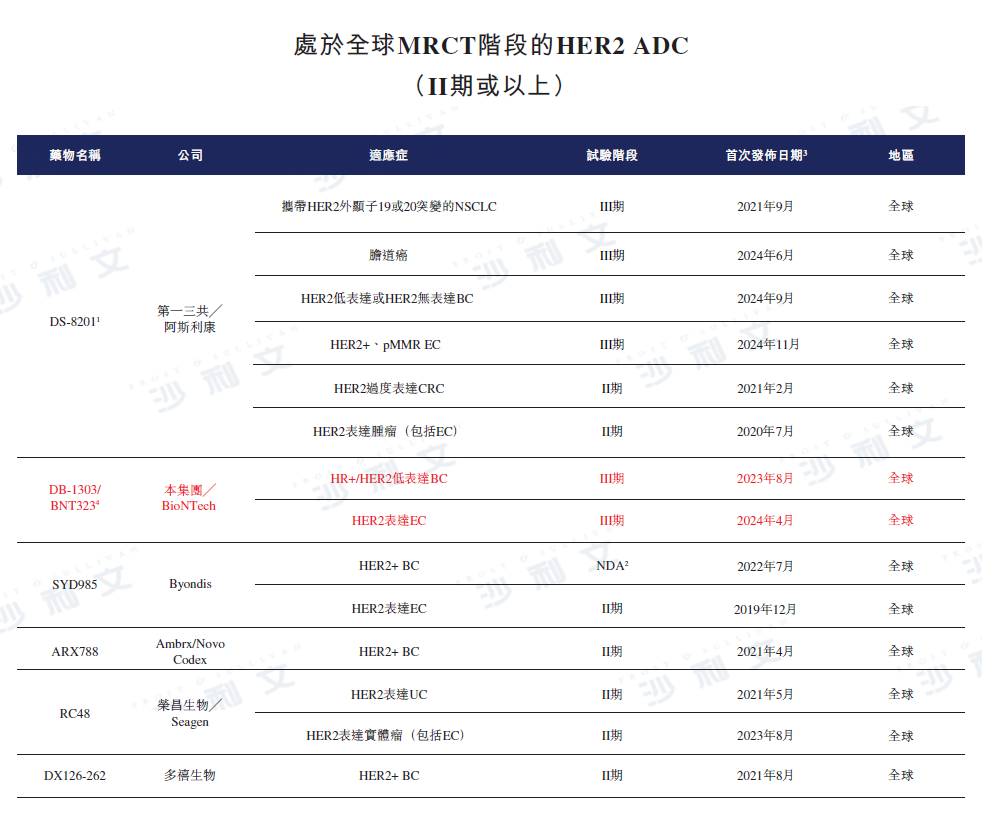
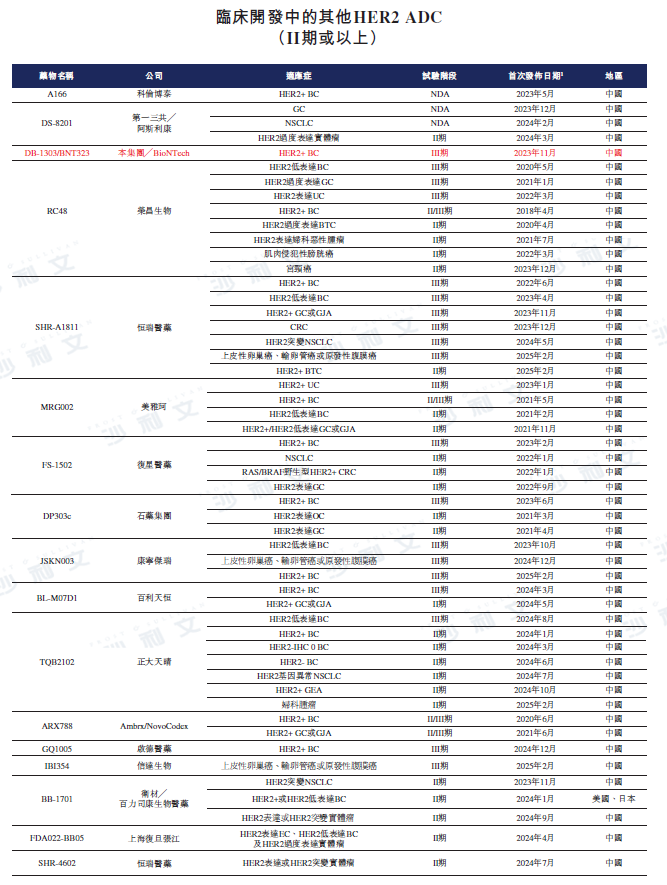
Data source: Clinicaltrials.gov, Center for Drug Evaluation, Frost & Sullivan analysis
Frost & Sullivan, integrating 64 years of global consulting experience, has dedicated 27 years to serving the booming Chinese market. With a global perspective, it helps clients accelerate their business growth, achieving benchmark positions in industry growth, innovation, and leadership. The healthcare industry is one of Frost & Sullivan's core areas of focus. Over the past 20-plus years, the Frost & Sullivan team has provided financing, financial advisory, IPO industry advisory, strategic consulting, and management consulting services to hundreds of outstanding domestic and international biopharmaceutical, medical device, healthcare services, and internet healthcare companies. Successful listings include: Weisun Pharmaceutical (2561.HK), Yasheng Pharmaceutical Group (NASDAQ:AAPG), Zhengye Biotech (NASDAQ:ZYBT), BrainGate (6681.HK), Health Road (2587.HK), Huahao Zhongtian (2563.HK), Yinos (688710.SH), Jingtai Technology (2228.HK), Yimai Sunshine (2522.HK), Shenghe Biotech (2898.HK), Quanxin Biotech (2509.HK), Meizhong Jiahé (2453.HK), WuXi AppTec (2268.HK), Anol Medical (NASDAQ:ANL), Neusoft Xikang (9686.HK), Youzhiyou (2496.HK), Yiming Angke (1541.HK), Cerenbo Tai (6990.HK), LaiKai Medical (2105.HK), Cedi Group (2487.HK), Lvt Bio (2480.HK), Meis Health (2415.HK), PHECR, Zhongjin Medical (NASDAQ:ZJYL), Yisheng Biotech (NASDAQ:YS), Meili Tianyuan (2373.HK), Kangfeng Biotech (6922.HK), Bao'an Biotech (6955.HK), Sida Di (1244.HK), Meihao Medical (1947.HK), Gaoshi Medical (2407.HK), Lepu Xin Tai (2291.HK), Jian Shi Technology (9877.HK), Health Yuan (JCARE.SW), Lepu Medical (LEPU.SW), Dingdang Health (9886.HK), Bio-Oncology (2315.HK), Zhiyun Health (9955.HK), MeinGene (6667.HK), Prenetics (PRE.NASDAQ), Yunkang Group (2325.HK), Ruike Biotech (2179.HK), Lepu Biotech (2157.HK), Clear Medical (1406.HK), Baxin An (2185.HK), Yonghe Medical (2279.HK), Kailai Ying (6821.HK), Beihai Kangcheng (1228.HK), Gusheng Tang (2273.HK), Yingpeng Technology (2251.HK), Clover Biotech (2197.HK), Minimally Invasive Robotics (2252.HK), Harmony Kamman (2256.HK), Kunbo Medical (2216.HK), Xianruida (6669.HK), Kangsheng Global (9960.HK), Yimai Tong (2192.HK), Tengsheng BOP (2137.HK), Canola (2162.HK), Chaoyu Eye Hospital (2219.HK), Guichuang Tongqiao (2190.HK), Hehuang Medicine (0013.HK), Koji Pharmaceutical (2171.HK), Zhaoke Eye Hospital (6622.HK), Nature's Pharmacy (UPC.NASDAQ), Sainfo Pharmaceutical (6600.HK), Zhaoyan New Drugs (6127.HK), Novogene Health (6606.HK), ADAG.NASDAQ, Beikang Medical (2170.HK), Jianbimiao Miao (2161.HK), Minimally Invasive Xin Tong (2160.HK), Rui Li Medical Beauty (2135.HK), Jiaosisi Pharmaceutical (1167.HK), Hebo Medicine (2142.HK), JD Health (6618.HK), Deqi Medicine (6996.HK), Rongchang Biotech (9995.HK), WuXi AppTec (2126.HK), SinoBIO (2096.HK), Yunding Newray (1952.HK), Jiahe Biotech (6998.HK), Zai Ding Medicine (9688.HK), Ou Kang Weishi (1477.HK), Yongtai Biotech (6978.HK), Haipu Pharmaceutical (9989.HK), Kepai Pharmaceutical (9939.HK), Peijia Medical (9996.HK), Kangfang Biotech (9926.HK), Nuo Cheng Jianhua (9969.HK), Tianjing Biotech (IMAB.NASDAQ), Kanglong Chemical (3759.HK), China Antibody (3681.HK), Dongyao Pharmaceutical (1875.HK), Yasheng Pharmaceutical (6855.HK), Fuhong Hanlin (2696.HK), Hansoh Pharmaceutical (3692.HK), Mabotech (2181.HK), Fangda Holdings (1521.HK), Via Bio (1873.HK), CStone Pharmaceuticals (2616.HK), Junshi Biotech (1877.HK), WuXi AppTec (2359.HK), Xinda Biotech (1801.HK), Hualing Medicine (2552.HK), BeiGene (6160.HK), Gilead Sciences (1672.HK), WuXi Biotech (2269.HK), China Resources Medicine (3320.HK), Yacogen Scientific & Pharmaceutical (2633.HK), HCM.NASDAQ, Biotech (1548.HK), BBI Life Sciences (1035.HK), Tongyuan Kang Medicine (2410.HK), etc. In terms of the number of filings, the Frost & Sullivan healthcare team maintains an absolute leading position in Hong Kong healthcare IPOs, consistently ranking first in market share from 2018 to 2023.
Since the listing of the first batch of companies on the Sci-tech Innovation Board in July 2019, Frost & Sullivan reports have been widely cited in the prospectuses of leading Sci-tech Innovation Board listed companies in the industry. These include: Zhongyan Co., Ltd. (688716.SH), Zhejiang Rongtai (603119.SH), Optoelectronics Technology Group Co., Ltd. (688450.SH), Jinghe Integration (688249.SH), Wuxi Rilian (688531.SH), Maolai Optical (688502.SH), Kangwe Century (688426.SH), Jinchuan Protein (688137.SH), Novogene Biologics (688428.SH), Aopu Mabio (688293.SH), MicroPort Electrophysiology (688351.SH), Mengke Pharmaceutical (688373.SH), Yifang Biologics (688382.SH), Jicui Pharmaceutical (688046.SH), Haichuang Pharmaceutical (688302.SH), Rongchang Biologics (688331.SH), Rendu Biologics (688193.SH), Shouyao Holdings (688197.SH), Heyuan Biologics (688238.SH), Yaxin Security (688225.SH), Xidiwei (688173.SH), Maiwei Biologics (688062.SH), Yahong Medicine (688176.SH), BeiGene (688235.SH), Jiahe Meikang (688246.SH), Dizhe Medicine (688192.SH), Novozyme (688105.SH), Chengda Biologics (688739.SH), Geke Micro (688728.SH), Huaxi Biologics (688363.SH), Junshi Biologics (688180.SH), Zhejiang Oncology Sciences (688266.SH), Beiogen (688177.SH), Shenzhou Cells (688520.SH), etc., are considered to be one of the most powerful, professional, and influential industry research institutions in the sector. We hope to work with enterprises to understand industry trends, seize development opportunities, jointly promote innovation and upgrading of China's health industry, and build a healthy future.
Recommended Reads (scroll up and down for more)
Frost & Sullivan assists Zhengye Biology in successfully going public in the US (NASDAQ:ZYBT)
Frost & Sullivan helps Health Road successfully list on the Hong Kong Stock Exchange (2587.HK)
Frost & Sullivan assisted Yipui Sunshine to successfully go public in Hong Kong (2522.HK)
Frost & Sullivan assists Tsuen Shin Bio to successfully go public in Hong Kong (2509.HK)
Frost & Sullivan Eastsoft success to hong listed(9686.HK)
Frost & Sullivan helps Yiming Angke successfully go public in Hong Kong (1541.HK)
Frost & Sullivan assists Lüzhoubio in successfully listing on the Hong Kong Stock Exchange (2480.HK)
Frost & Sullivan helps MesHealth successfully go public in Hong Kong (2415.HK)
Frost & Sullivan assists Zhongjin Medical in successfully going public in the US (NASDAQ: ZJYL)
Frost & Sullivan assisted Yisheng Biology in successfully going public on the NASDAQ (NASDAQ:YS)
Frost & Sullivan assists Meiliyuan in successfully listing on the Hong Kong Stock Exchange (2373.HK)
Frost & Sullivan helps Thundersoft successfully go public in Hong Kong (1244.HK)
Frost & Sullivan helps Lepuventec successfully go public in Hong Kong (2291.HK)
Frost & Sullivan helps Bio-TheraPlex successfully list on the Hong Kong Stock Exchange (2315.HK)
Frost & Sullivan helps Zhiyun Health successfully go public in Hong Kong (9955.HK)
Frost & Sullivan assists MeinGene in successfully listing on the Hong Kong Stock Exchange (6667.HK)
Frost & Sullivan assists Prenetics in successfully going public on the NASDAQ (NASDAQ:PRE)
Frost & Sullivan assists Clear Medical in successfully going public in Hong Kong (1406.HK)
Frost & Sullivan assists Centric HeartCare in successfully going public in Hong Kong (2185.HK)
Frost & Sullivan assists Kaleido in successfully listing on the Hong Kong Stock Exchange (6821.HK)
Frost & Sullivan helps Gushengtang successfully go public in Hong Kong (2273.HK)
Frost & Sullivan assisted Yuhuan in successfully listing on the Hong Kong Stock Exchange (2256.HK)
Frost & Sullivan assists Kunbo Medical in successfully going public in Hong Kong (2216.HK)
Frost & Sullivan helps CanSino Biologics successfully go public in Hong Kong (2162.HK)
Frost & Sullivan helps Chaoyu Eye Hospital successfully go public in Hong Kong (2219.HK)
Frost & Sullivan helps Guichuang Tongqiao successfully go public in Hong Kong (2190.HK)
Frost & Sullivan helps Huarong Medicine successfully go public in Hong Kong (0013.HK)
Frost & Sullivan helps Kite Pharma successfully go public in Hong Kong (2171.HK)
Frost & Sullivan helps Zhaoke Eye Hospital successfully go public in Hong Kong (6622.HK)
Frost & Sullivan helps Nature Medicine successfully go public in the US (NASDAQ):UPC)
Frost & Sullivan helps Sino Biologics successfully go public in Hong Kong (6600.HK)
Frost & Sullivan helps Zhaoyan New Drugs successfully go public in Hong Kong (6127.HK)
Frost & Sullivan helps Novartis Health successfully go public in Hong Kong (6606.HK)
Frost & Sullivan helps Tianyan Pharmaceutical successfully go public in the US (NASDAQ:ADAG)
Frost & Sullivan helps Beicang Medical successfully go public in Hong Kong (2170.HK)
Frost & Sullivan helps Jianbimiao successfully go public in Hong Kong (2161.HK)
Frost & Sullivan helps Rui Li Medical Beauty successfully go public in Hong Kong (2135.HK)
Frost & Sullivan helps Jiake Pharmaceutical successfully go public in Hong Kong (1167.HK)
Frost & Sullivan helps Hengbo Medicine successfully go public in Hong Kong (2142.HK)
Frost & Sullivan helps JD Health successfully go public in Hong Kong (6618.HK)
Frost & Sullivan helps Deqi Medicine successfully go public in Hong Kong (6996.HK)
Frost & Sullivan helps Rongchang Biotech successfully go public in Hong Kong (9995.HK)
Frost & Sullivan helps WuXi AppTec successfully go public in Hong Kong (2126.HK)
Frost & Sullivan helps First Voice Pharmaceuticals successfully go public in Hong Kong (2096.HK)
Frost & Sullivan helps Yunding New Horizon successfully go public in Hong Kong (1952.HK)
Frost & Sullivan helps Jiahe Biotech successfully go public in Hong Kong (6998.HK)
Frost & Sullivan helps Zai Lab successfully go public in Hong Kong (9688.HK)
Frost & Sullivan helps OcularVista successfully go public in Hong Kong (1477.HK)
Frost & Sullivan helps Yongtai Biotech successfully go public in Hong Kong (6978.HK)
Frost & Sullivan helps Hapreya Pharmaceuticals successfully go public in Hong Kong (9989.HK)
Frost & Sullivan helps Pioneering Pharmaceuticals successfully go public in Hong Kong (9939.HK)
Frost & Sullivan helps Peijia Medical successfully go public in Hong Kong (9996.HK)
Frost & Sullivan helps Kangfang Biotech successfully go public in Hong Kong (9926.HK)
Frost & Sullivan helps Nuo Cheng Jian Hua successfully go public in Hong Kong (9969.HK)
Frost & Sullivan helps Tianjing Biotech successfully go public in the US (NASDAQ:IMAB)
Frost & Sullivan helps Concha Biologics successfully go public in Hong Kong (3759.HK)
Frost & Sullivan helps China Antibody successfully go public in Hong Kong (3681.HK)
Frost & Sullivan helps Dongyao Pharmaceuticals successfully go public in Hong Kong (1875.HK)
Frost & Sullivan helps Yasheng Medicine successfully go public in Hong Kong (6855.HK)
Frost & Sullivan helps Fosun Pharma successfully go public in Hong Kong (2696.HK)
Frost & Sullivan helps Hansoh Pharmaceutical successfully go public in Hong Kong (3692.HK)
Frost & Sullivan helps Mabtech Pharmaceuticals successfully go public in Hong Kong (2181.HK)
Frost & Sullivan helps Fangda Holdings successfully go public in Hong Kong (1521.HK)
Frost & Sullivan helps ViaBio successfully go public in Hong Kong (1873.HK)
Frost & Sullivan helps CStone Pharmaceuticals successfully go public in Hong Kong (2616.HK)
Frost & Sullivan helps Junshi Biosciences successfully go public in Hong Kong (1877.HK)
Frost & Sullivan helps WuXi AppTec successfully go public in Hong Kong (2359.HK)
Frost & Sullivan helps Xinda Biotech successfully go public in Hong Kong (1801.HK)
Frost & Sullivan helps Valgen Pharmaceuticals successfully go public in Hong Kong (2552.HK)
Frost & Sullivan helps BeiGene successfully go public in Hong Kong (6160.HK)
Frost & Sullivan helps Gilead Sciences successfully go public in Hong Kong (1672.HK)
Frost & Sullivan helps WuXi AppTec Biologics successfully go public in Hong Kong (2269.HK)
Frost & Sullivan helps China Resources Pharma successfully go public in Hong Kong (3320.HK)
Frost & Sullivan helps Yakult Biopharmaceuticals successfully go public in Hong Kong (2633.HK)
Frost & Sullivan helps Huarong China Medicine successfully go public in the US (NASDAQ:HCM)
Frost & Sullivan helps Kingsbridge Biosciences successfully go public in Hong Kong (1548.HK)
Frost & Sullivan helps BBI Life Sciences successfully go public in Hong Kong (1035.HK)
Frost & Sullivan helps Tongyuan Kang Medicine successfully go public in Hong Kong (2410.HK)
*The above order is not in any particular sequence and is arranged in reverse order of listing time

