Beijing Green Bamboo Biotechnology Co., Ltd. (Stock Code: 02480.HK) successfully listed on the main board of the Hong Kong capital market on May 8, 2023. The company is a biotechnology company dedicated to providing high-quality and affordable human vaccines and therapeutic biological agents to prevent and control infectious diseases, treat cancer, and meet unmet clinical needs for autoimmune diseases. After more than two decades of independent research and technological innovation, the company has developed Fabite®The technology platform and mammalian expression technology platform have laid a solid foundation for the development of human vaccines, monoclonal antibody products, and bispecific antibody research products. The company's current product pipeline includes three clinical-stage products, including the core products LZ901, K193, and K3, as well as four preclinical products. Among them, the core product LZ901 is a recombinant herpes zoster vaccine independently developed by the company and is expected to become the world's first herpes zoster vaccine with a tetrameric molecular structure. Frost & Sullivan (hereinafter referred to as 'Frost & Sullivan') provides exclusive industry advisory services for the listing of Beijing Lvx Biotec Co., Ltd., and hereby warmly congratulates it on its successful listing.

Beijing Green Bamboo Biotechnology Co., Ltd. (hereinafter referred to as 'Green Bamboo Biotech') successfully went public on May 8, 2023. The company issued a total of 1,038.6 million shares globally, including 103.88 million shares in Hong Kong and 9.3472 billion shares internationally. The issue price was HK$32.80, raising approximately HK$241 million net proceeds.
During the Hong Kong listing process, Frost & Sullivan mainly undertook the following tasks: helping the issuer accurately and objectively understand its positioning in the target market, using objective market data to discover, support, and highlight the issuer's competitive advantages, assisting the issuer, sponsor, and other professional intermediary institutions in completing the writing of relevant parts of the prospectus (such as the overview, competitive advantages and strategy, industry overview, business, and other important sections), facilitating communication between the issuer and the Hong Kong Stock Exchange and investors, helping investors quickly understand the market ecosystem and competitive landscape, and assisting the issuer in completing feedback on industry-related issues from the Hong Kong Stock Exchange.
Investment highlights
Currently, GSK's Shingrix®The zoster live attenuated vaccine developed by Bihaike Biotechnology is the only two zoster vaccines approved for marketing in China.
The company's LZ901 is expected to become China's first domestically developed recombinant herpes zoster vaccine. In the clinical Phase I trial, LZ901 was compared with Shingrix®A head-to-head trial was conducted, showing better safety;
The company's R&D team has been deeply involved in the research and development of innovative vaccines and antibody drugs for over two decades and possesses rich R&D experience;
The company has established multiple innovative technology platforms, capable of developing recombinant vaccines and antibody products with improved efficiency, purity, and stability.
According to a report by Frost & Sullivan:
The global herpes zoster vaccine market size is expected to grow from $2.4 billion in 2021 to reach $12.6 billion by 2030;
LZ901 is a recombinant zoster vaccine independently developed by the company. It is expected to become the world's first zoster vaccine with a tetrameric molecular structure, intended for the prevention of herpes zoster caused by varicella-zoster virus (VZV) in adults aged 50 and above.
K193 is an investigational product of the company's own development, a bispecific antibody injection for the treatment of B-cell leukemia and lymphoma. Currently, there is only one bispecific antibody injection (CD19-CD3) approved in China.
Global Herpes Zoster Vaccine Market Overview
Herpes zoster is a disease caused by the varicella-zoster virus and is highly contagious. Once a person has had chickenpox as a child, the virus remains in their body and may reactivate as herpes zoster years later. Middle-aged and elderly people over the age of 50 are at high risk for herpes zoster. According to the recommendations of the US FDA, vaccination with the herpes zoster vaccine can effectively prevent herpes zoster and postherpetic neuralgia. As of now, there are four types of herpes zoster vaccines available on the market globally.
Based on sales revenue, the global herpes zoster vaccine market increased from $700 million in 2015 to $24 billion in 2021, with a compound annual growth rate of 21.2%. It is expected to grow to $61 billion by 2025. The global herpes zoster vaccine market is expected to increase to $126 billion by 2030.
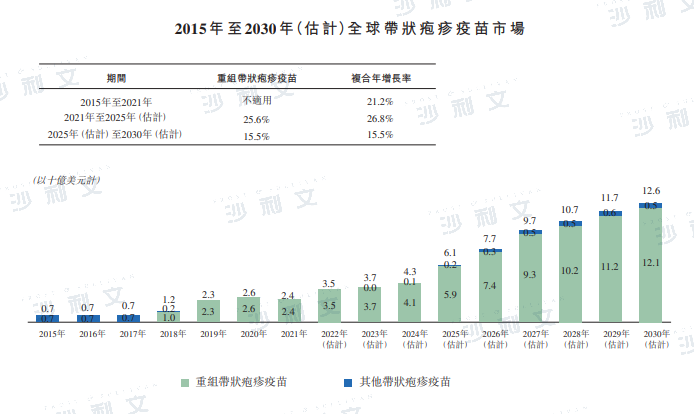
Data source: Public disclosures by listed companies, expert interviews, Frost & Sullivan analysis
According to the '2022 Expert Consensus on Herpes Zoster Vaccination in China', it is recommended that people aged 50 and above receive the herpes zoster vaccine for prevention. With the public's increasing awareness of herpes zoster, it is expected that the market for herpes zoster vaccines in China will grow significantly. In terms of sales revenue, the Chinese herpes zoster vaccine market is projected to increase from RMB 600 million in 2021, at a compound annual growth rate of 103.8%, to RMB 108 billion by 2025, and further to RMB 281 billion by 2030.
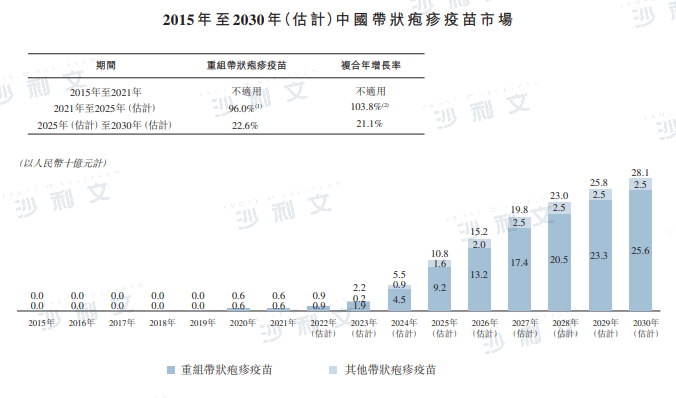
Data source: Expert interviews, Frost & Sullivan analysis
Key drivers
The growth drivers of the global herpes zoster vaccine market include (i) ongoing population aging; (ii) a continuous increase in the number of new herpes zoster cases; and (iii) the lack of effective treatment.
-
Sustained population aging
With the decline in fertility and the extension of average life expectancy, the population aged 50 and above is expected to continue growing in the future. By 2030, the global population aged 50 and above is expected to reach 2.33 billion, accounting for about 27.3% of the total global population. In 2030, China's population aged 50 and above is expected to reach 570 million, accounting for about 39.0% of China's total population. The decline in the immune ability of people over 50 years old makes them a high-risk group for shingles.
-
The number of new cases of herpes zoster continues to grow
In 2021, among the population aged 50 and above, new cases of herpes zoster reached 3.9 million in China, 1.1 million in the United States, and 2.0 million in Europe, respectively. Since the number of new herpes zoster cases is still on the rise in the future, and currently China's vaccination rate against herpes zoster is lower compared to European and American countries, it is expected that there will be significant room for growth in China's herpes zoster vaccination rate in the future.
-
Lack of effective treatment methods
Herpes zoster, accompanied by complications such as neuralgia, can affect sleep, mood, work, and daily life. According to China's 2018 clinical guidelines for herpes zoster, there are currently no specific medications for the condition. Vaccination can prevent herpes zoster and greatly reduce the burden of related diseases.
competitive landscape
As of the latest practicable date, a total of four herpes zoster vaccines have been launched globally, including Merck's Zostavax.®Glenox's Shingrix®SK Chemical's SkyZoster®And Biorad's VaxImmune. Among them, Zostavax®Due to low effectiveness and weak market competitiveness, it was discontinued in the United States in November 2020; SkyZoster®Mainly sold in South Korea; Bial Bio's varicella-zoster vaccine, CanSinoVac, was approved for marketing in January 2023 and is only available in China.
LZ901 is the first herpes zoster vaccine to be submitted for clinical trials in both China and the United States. Based on the immunogenicity data from the Phase I clinical trial, the level of anti-VZV antibodies after a full course of vaccination with LZ901 is comparable to that of Shingrix.®There is no significant difference compared to Shingrix.®In comparison, the incidence of adverse reactions related to the aluminum hydroxide adjuvant used in LZ901 is expected to be lower than that of Shingrix.®An oily adjuvant used in China. LZ901 provides strong protection against herpes zoster.
The following table lists the details of herpes zoster vaccines under development in China, the United States, and other countries:
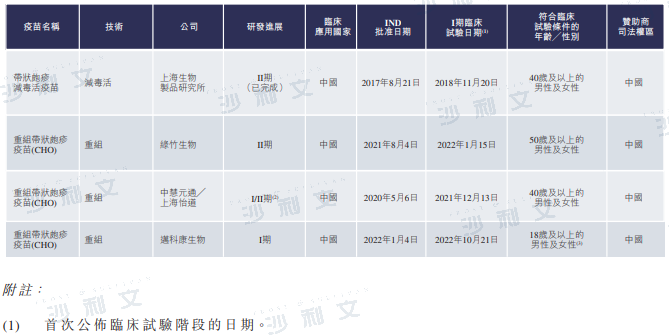
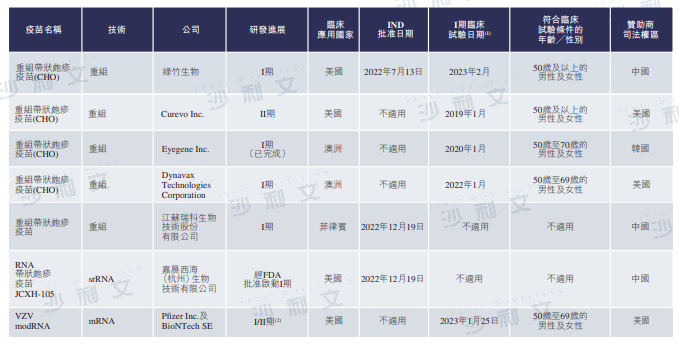
Source: CDE, published disclosure, Frost & Sullivan analysis
Overview of the Recurrent and Refractory B-cell Non-Hodgkin Lymphoma (NHL)/Acute Lymphoblastic Leukemia (ALL) Treatment Market
The incidence of malignant tumors of the hematopoietic system such as lymphoma, myeloma, and leukemia is increasing year by year. Although the application of various innovative drugs and therapies has greatly improved clinical treatment outcomes, the challenge of refractory and difficult-to-treat hematopoietic system tumors remains prominent. Lymphoma is a malignant tumor originating from the lymphatic hematopoietic system and is the most common blood tumor worldwide.
Among them, non-Hodgkin lymphoma (NHL) is the main category in lymphoma classification, accounting for 90%. Most clinical NHL cases are B-cell types, making up 70% to 85% of the total. In 2021, the number of new cases of NHL in China reached 95,000. Acute lymphoblastic leukemia (ALL) is a heterogeneous hematological malignancy, with 80% occurring in children. In 2021, the number of new cases of ALL in China was about 13,000.
Currently, the main treatment methods for relapsed and refractory B-cell NHL and B-cell ALL include CD3/CD19 bispecific antibodies, CD19 antibody conjugates (ADCs), CAR-T cell therapy, and hematopoietic stem cell transplantation. The market size of the relapsed and refractory B-cell NHL/ALL treatment market in China reached 269 billion yuan in 2021. With the continuous introduction of innovative therapies, it is expected that the market size will further grow to 357 billion yuan by 2030.
The following table lists the historical and future forecasts of the market size for the treatment of relapsed/refractory B-cell NHL/ALL in China:
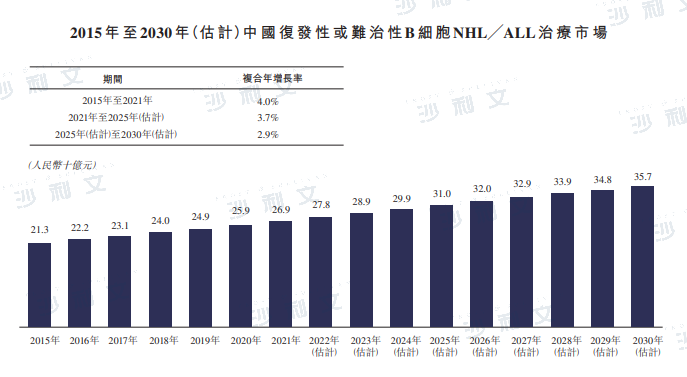
Data source: Analysis by Frost & Sullivan
growth drivers
(i) Currently, there are few treatment options for relapsed and refractory B-cell NHL/ALL, which is an unmet clinical need. Patients with relapsed and refractory B-cell NHL/ALL are those who have failed first-line treatment and require continuation with second or third-line therapy. According to the 2021 CSCO Guidelines for the Diagnosis and Treatment of Lymphoma and the 2021 CSCO Guidelines for the Diagnosis and Treatment of Hematologic Malignancies, there are no specific drugs for patients with relapsed and refractory B-cell NHL/ALL.
(ii) Due to limited options for mechanism of action, existing therapies cannot provide effective treatment for patients with relapsed or refractory diffuse large B-cell lymphoma. For example, rituximab maintenance therapy has no significant effect on relapsed or refractory diffuse large B-cell lymphoma after autologous stem cell transplantation. Compared with the United States, China also has extremely limited second-line treatment options other than chemotherapy.
(iii) The diagnosis of hematological malignancies is usually made through bone marrow examination and imaging. Most patients choose chemotherapy, targeted drugs, immunotherapy, and for those who are eligible, bone marrow transplantation. The main shortcomings of current therapies include low overall response rates, high recurrence rates, significant side effects, long treatment cycles, and high costs. Due to the lack of key drugs, insufficient adjuvant therapy, and low early diagnosis rates, the five-year survival rate for hematological malignancies in China is very low, with the five-year survival rates for NHL and multiple myeloma being lower than those for the same indications in the United States.
(iv) The Chinese government is implementing favorable policies and regulations to promote the development of innovative therapies. Special review channels such as priority review have also accelerated the market launch of anti-tumor drugs for conditions like hematological malignancies. At the same time, policies such as expanding medical insurance coverage, zero tariffs on imported anti-cancer drugs, and inclusion of anti-cancer drugs in medical insurance negotiations will further reduce the cost of anti-cancer drugs and further improve the accessibility of innovative tumor immunotherapy drugs.
competitive landscape
As of the latest practicable date, there are three marketed drugs in China for the end-of-line treatment of relapsed and refractory B-cell NHL/ALL, including a bispecific antibody and two CAR-T cell therapies.
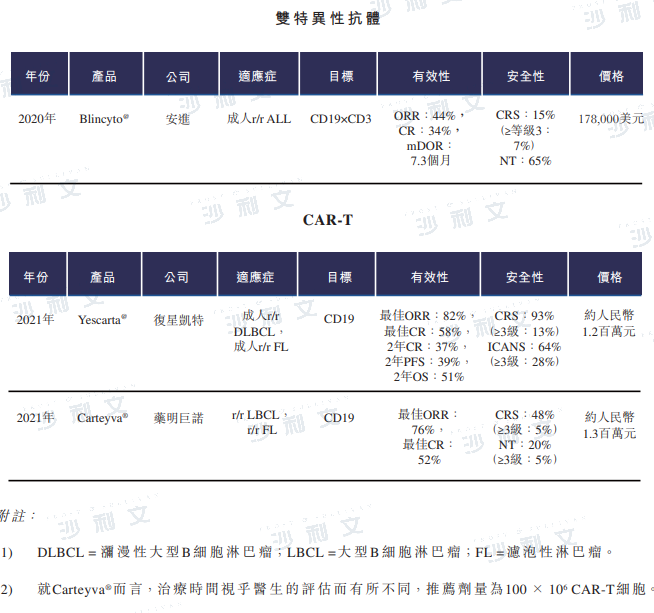
Data source: Guangdong Pharmaceutical Electronic Trading Platform, Frost & Sullivan analysis
As of the latest practicable date, China has one CD3/CD19 bispecific antibody that is already on the market for the treatment of relapsed/refractory B-cell NHL/ALL, five bispecific antibodies in development, and one CD3/CD19/CD20 trispecific antibody.
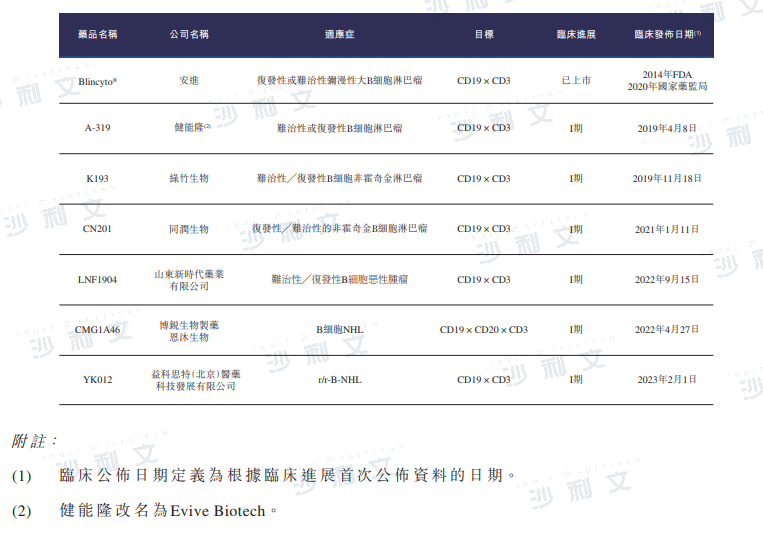
Data source: Analysis by Frost & Sullivan
Click at the end of the articleRead the original textView the prospectus
Frost & Sullivan, integrating 62 years of global consulting experience, has dedicated 25 years to serving the booming Chinese market with a global perspective, helping clients accelerate their corporate growth and achieve benchmark positions in industry growth, innovation, and leadership. The healthcare industry is one of the core areas of focus for Frost & Sullivan. Over the past 20+ years, the Frost & Sullivan team has provided financing financial advisory, IPO industry advisory, strategic consulting, management consulting, and other services to hundreds of outstanding domestic and international biopharmaceuticals, medical devices, healthcare services, and internet healthcare companies. Successful listings include: Meis Health (2415.HK), PHECR, ZJYL, YS, Meiliyuan (2373.HK), Kangfeng Biotech (6922.HK), Bao'an Biotech (6955.HK), Sididi (1244.HK), Meihao Medical (1947.HK), Gaoshi Medical (2407.HK), Lepu Xin Tai (2291.HK), Jian Shi Technology (9877.HK), Health Yuan (JCARE.SW), Lepu Medical (LEPU.SW), Dingdang Health (9886.HK), Bao'ao Saitu (2315.HK), Zhiyun Health (9955.HK), Mein Gene (6667.HK), Prenetics (PRE.NASDAQ), Yunkang Group (2325.HK), Rike Biotech (2179.HK), Lepu Biotech (2157.HK), Clear Medical (1406.HK), Baxin An (2185.HK), Yonghe Medical (2279.HK), Kailai Ying (6821.HK), Beihai Kangcheng (1228.HK), Gusheng Tang (2273.HK), Yingtong Technology (2251.HK), Clover Biotech (2197.HK), Minimally Invasive Robotics (2252.HK), Harmony Cayman (2256.HK), Kunbo Medical (2216.HK), Xianruida (6669.HK), Kangsheng Global (9960.HK), Yimai Tong (2192.HK), Tengsheng Bovy (2137.HK), CanSino (2162.HK), Chaoyuju Eye Hospital (2219.HK), Guichuang Tongqiao (2190.HK), Huihuang Medicine (0013.HK), Koi Pharmaceutical (2171.HK), Zhaoke Eye Hospital (6622.HK), Nature Medicine (UPC.NASDAQ), Saiseng Pharmaceutical (6600.HK), Zhaoyan New Drugs (6127.HK), Novogene Health (6606.HK), ADAG.NASDAQ, Beikang Medical (2170.HK), Jianbimiao Miao (2161.HK), Minimally Invasive Xin Tong (2160.HK), Rui Li Medical Beauty (2135.HK), Jiake Pharmaceutical (1167.HK), Hepatox (2142.HK), JD Health (6618.HK), Deqi Pharmaceutical (6996.HK), Rongchang Biotech (9995.HK), WuXi AppTec (2126.HK), Sino Biologics (2096.HK), Yunding New Energy (1952.HK), Jiahe Biotech (6998.HK), Zai Ding Pharmaceutical (9688.HK), Ocular Biotech (1477.HK), Yongtai Biotech (6978.HK), Haipu Pharmaceutical (9989.HK), Kaidao Pharmaceutical (9939.HK), Peijia Medical (9996.HK), Kangfang Biotech (9926.HK), Nuo Cheng Jianhua (9969.HK), Tianjing Biotech (IMAB.NASDAQ), Kanglong Chemical (3759.HK), China Antibody (3681.HK), Dongyao Pharmaceutical (1875.HK), Yasheng Pharmaceutical (6855.HK), Fuhong Hanlin (2696.HK), Hansoh Pharmaceutical (3692.HK), Mabotech (2181.HK), Fangda Holdings (1521.HK), Via Biotech (1873.HK), CStone Pharmaceuticals (2616.HK), Junshi Biosciences (1877.HK), WuXi AppTec (2359.HK), Innovent Biologics (1801.HK), Hailun Medicine (2552.HK), BeiGene (6160.HK), Galle Therapeutics (1672.HK), WuXi AppTec (2269.HK), China Resources Medicine (3320.HK), Yakuten Scientific Research Pharmaceutical (2633.HK), Huihuang China Medicine (HCM.NASDAQ), Genscript Biotechnology (1548.HK), BBI Life Sciences (1035.HK), etc. In terms of the number of filings, the Frost & Sullivan healthcare team maintains an absolute leading position in Hong Kong's healthcare IPOs, consistently ranking first in market share from 2018 to 2022.
Since the listing of the first batch of companies on the Sci-tech Innovation Board in July 2019, Frost & Sullivan reports have been widely cited in the prospectuses of leading Sci-tech Innovation Board listed companies in the industry, including: Kangwei Century (688426.SH), Jinnan Protein (688137.SH), Nuo Cheng Jianhua (688428.SH), Aopu Mai Biotech (688293.SH), MicroPort Electrophysiology (688351.SH), Mengke Pharmaceutical (688373.SH), Yifang Biotech (688382.SH), Jicui Yaokang (688046.SH), Haichuang Pharmaceutical (688302.SH), Rongchang Biotech (688331.SH), Rendu Biotech (688193.SH), Shouyao Holdings (688197.SH), Heyuan Biotech (688238.SH), Yaxin Security (688225.SH), Xidi Micro (688173.SH), Mawei Biotech (688062.SH), Yahong Medicine (688176.SH), BeiGene (688235.SH), Jiahe Meikang (688246.SH), Dizhe Medicine (688192.SH), Novogene (688105.SH), Chengda Biotech (688739.SH), Geke Micro (688728.SH), Huaxi Biotech (688363.SH), Junshi Biotech (688180.SH), Zhejiang Genomics & Biotechnology Co., Ltd. (688266.SH), BeiGene (688177.SH), Shenzhou Cells (688520.SH), etc. They are considered one of the most powerful, professional, and influential industry research institutions in the sector. We hope to work with enterprises to understand industry trends, seize development opportunities, jointly promote innovation and upgrading of China's healthcare industry, and build a healthy future.
Recommended Reading
Frost & Sullivan assists MesHealth in successfully listing on the Hong Kong Stock Exchange (2415.HK)
Frost & Sullivan assists Zhongjin Medical in successfully going public in the US (NASDAQ: ZJYL)
Frost & Sullivan assisted Yisheng Biology in successfully going public on the NASDAQ (NASDAQ:YS)
Frost & Sullivan assists Meiliyuan in successfully listing on the Hong Kong Stock Exchange (2373.HK)
Frost & Sullivan assisted IdeaChip to successfully go public in Hong Kong (1244.HK)
Frost & Sullivan helps Lepuventec successfully go public in Hong Kong (2291.HK)
Frost & Sullivan assisted Health Yuan in successfully issuing GDRs and listing on the RSE (JCARE.SW)
Frost & Sullivan helps Dingdang Health successfully go public in Hong Kong (9886.HK)
Frost & Sullivan helps Bio-TheraPlex successfully list on the Hong Kong Stock Exchange (2315.HK)
Frost & Sullivan helps Zhiyun Health successfully go public in Hong Kong (9955.HK)
Frost & Sullivan assists MeinGene in successfully listing on the Hong Kong Stock Exchange (6667.HK)
Frost & Sullivan assisted Prenetics in successfully going public on the NASDAQ (NASDAQ:PRE)
Frost & Sullivan assists Clear Medical in successfully going public in Hong Kong (1406.HK)
Frost & Sullivan assists Baisin'an in successfully listing on the Hong Kong Stock Exchange (2185.HK)
Frost & Sullivan assists Kaleido in successfully listing on the Hong Kong Stock Exchange (6821.HK)
Frost & Sullivan helps Gushengtang successfully go public in Hong Kong (2273.HK)
Frost & Sullivan helps Minimally Invasive Robotics successfully go public in Hong Kong (2252.HK)
Frost & Sullivan assisted Yuhuan in successfully listing on the Hong Kong Stock Exchange (2256.HK)
Frost & Sullivan assists Kunbo Medical in successfully going public in Hong Kong (2216.HK)
Frost & Sullivan assists ConocoAL in successfully listing on the Hong Kong Stock Exchange (2162.HK)
Frost & Sullivan helps Guichuangtongqiao successfully go public in Hong Kong (2190.HK)
Frost & Sullivan assists Nature Pharma in successfully going public in the US (NASDAQ):UPC)
Frost & Sullivan helps Jianbaimiao Miao successfully go public in Hong Kong (2161.HK)
Frost & Sullivan helps JD Health successfully go public in Hong Kong (6618.HK)
Frost & Sullivan assists Oculent in successfully listing on the Hong Kong Stock Exchange (1477.HK)
Frost & Sullivan helps develop PharmaCure successfully go public in Hong Kong (9939.HK)
Frost & Sullivan assisted Tianjing BIO in successfully going public on the NASDAQ (NASDAQ: IMAB)
Frost & Sullivan assisted ViaBio in successfully listing on the Hong Kong Stock Exchange (1873.HK)
Frost & Sullivan helps WuXi AppTec successfully go public in Hong Kong (2359.HK)
Frost & Sullivan helps Cinda Biologics successfully list on the Hong Kong Stock Exchange (1801.HK)
Frost & Sullivan helps Hualing Medicine successfully list on the Hong Kong Stock Exchange (2552.HK)
Frost & Sullivan helps BeiGene successfully list on the Hong Kong Stock Exchange (6160.HK)
Frost & Sullivan helps Gilead Sciences successfully list on the Hong Kong Stock Exchange (1672.HK)
Frost & Sullivan helps WuXi Biologics successfully list on the Hong Kong Stock Exchange (2269.HK)
Frost & Sullivan helps Hutchison China Pharmaceutical successfully list on the US NASDAQ (HCM)
Frost & Sullivan helps King Biosciences successfully list on the Hong Kong Stock Exchange (1548.HK)
Frost & Sullivan helps BBI Life Sciences successfully list on the Hong Kong Stock Exchange (1035.HK)
*The above order is not chronological and is arranged in reverse order of listing

