
Lepu Xintai Medical Technology (Shanghai) Co., Ltd. (hereinafter referred to as 'Xintai Medical') successfully went public on November 8, 2022. The company plans to issue a total of 2,245,500 shares globally, including 2.246 million shares in Hong Kong and 2,020.9 million shares internationally, with an additional 15% over-allotment rights; the offering price is HK$29.15 per share.
During the process of listing in Hong Kong this time, Frost & Sullivan mainly undertook the following tasks: helping the issuer accurately and objectively understand its positioning in the target market, using objective market data to discover, support, and highlight the issuer's competitive advantages, assisting the issuer, investment banks, and other intermediaries in completing the relevant parts of the prospectus (such as overview, competitive advantages and strategy, industry overview, business, and other important chapters), facilitating communication between the issuer and the Exchange and investors, helping investors quickly understand the market ecosystem and competitive landscape, and assisting the issuer in completing feedback on various industry-related issues from the Exchange.
Investment highlights
-
The company has strong R&D capabilities and has taken the lead in investing in the research and development of biodegradable technologies;
-
The project 'Development and Clinical Application of Occluders and Related Devices for Interventional Treatment of Defective Congenital Heart Disease' has won the second prize for National Science and Technology Progress;
-
The company leads the research and development of biodegradable technologies, with such devices meeting various clinical treatment needs and being safe and effective;
-
The company has an advanced innovative spirit and industry-leading R&D capabilities.
According to the Frost & Sullivan report, as of 2021, the company:
-
Has 220 registered patents;
-
The leading enterprise in medical interventional devices for structural heart disease;
-
A pioneer in the development of biodegradable medical technology;
-
It is the largest manufacturer of congenital heart disease occluders and related surgical supporting products in China;
-
There are multiple commercially available and investigational occluders featuring biodegradable materials. These products have advanced to different pre-launch stages, including an occluder for the treatment of ventricular septal defect (a serious congenital heart disease), which was approved by the National Medical Products Administration in 2022 and is the first of its kind to be launched on the market.
Overview of the Medical Intervention Devices Market for Structural Heart Disease in China
Overview of the Market for Congenital Heart Disease Occluders in China
From 2017 to 2021, the market for congenital heart disease occluders in China expanded from 3.281 billion yuan to 4.264 billion yuan, with a compound annual growth rate of 6.8% during this period. The steady market growth stems from the expanding patient population and increasing affordability of medical services. It is estimated that by 2025, the market size for congenital heart disease occluders in China will reach 6.59 billion yuan, with a compound annual growth rate of 11.5% during the forecast period from 2021 to 2025. It is expected that by 2030, the market size will reach 7.658 billion yuan, with a compound annual growth rate of 3.1% during the forecast period from 2025 to 2030.
Market scale and forecast of congenital heart disease occluders in China
2017 to 2030 (estimated)
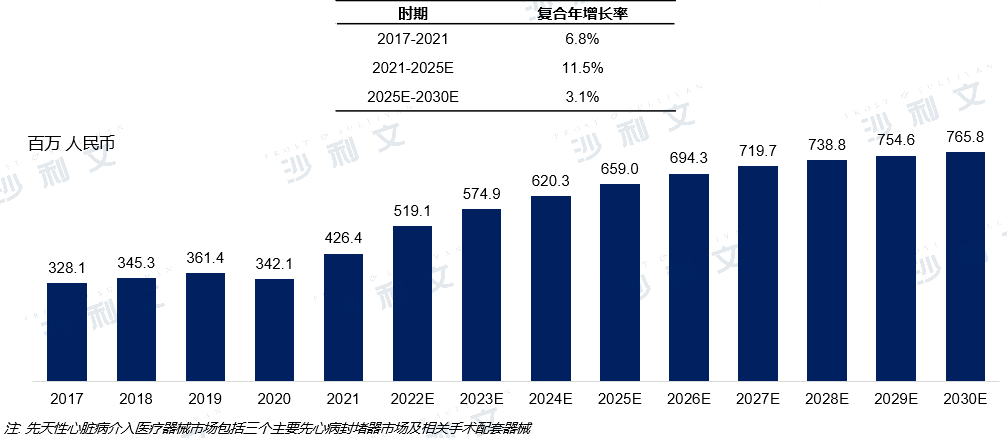
Data source: Analysis by Frost & Sullivan
Competitive landscape of the congenital heart disease occluder market in China
In China, domestic manufacturers of congenital heart disease occluders hold a dominant position in the market, accounting for about 91.5% of the market share in 2021, with the remaining about 8.5% occupied by international manufacturers. The following chart lists the top five participants in the Chinese occluder product market by revenue recognized from sales in 2021. The company is the largest manufacturer of congenital heart disease occluders and related surgical accessories in China.
The top five participants in the 2021 Chinese congenital heart disease occluder product market

Data source: Analysis by Frost & Sullivan
Market Overview of Atrial Septal Defect Occluders
The market scale of China's atrial septal defect occluders increased from 150 million yuan in 2017 to 210 million yuan in 2021, with a compound annual growth rate of 8.1%. The market scale will continue to expand, with an expected value of 290 million yuan by 2025 and 340 million yuan by 2030, with a compound annual growth rate of 3.2% from 2025 to 2030.
Market Scale and Forecast of China's Atrial Septal Defect Occluders
2017 to 2030 (estimated)
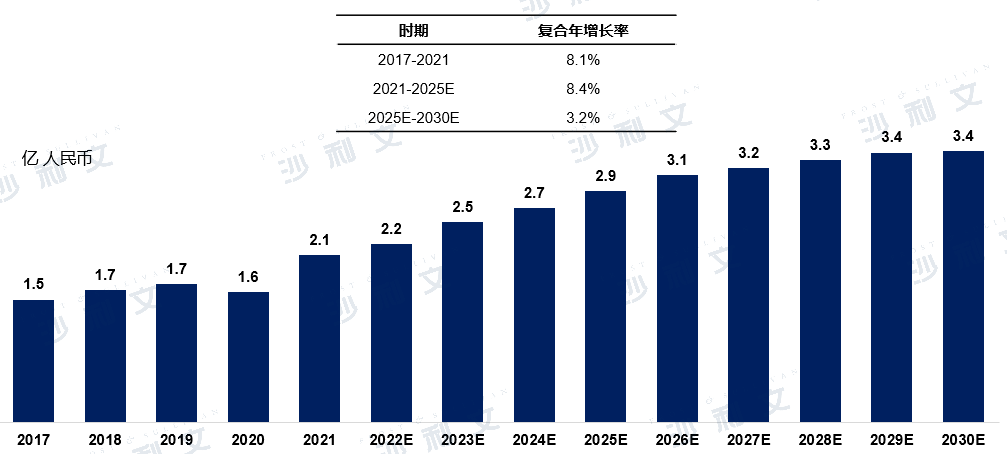
Data source: Analysis by Frost & Sullivan
Market Overview of Ventricular Septal Defect Occluders
The market scale of interventricular septal defect occluders in China increased from 46.8 million yuan in 2017 to 53 million yuan in 2021, with a compound annual growth rate of 3.1% from 2017 to 2021. It is estimated that by 2025, this figure will reach 73.4 million yuan, with a compound annual growth rate of 8.5% from 2021 to 2025. By 2030, the market scale is expected to reach 84.7 million yuan.
Market Scale and Forecast of Ventricular Septal Defect Occluders in China
2017 to 2030 (estimated)
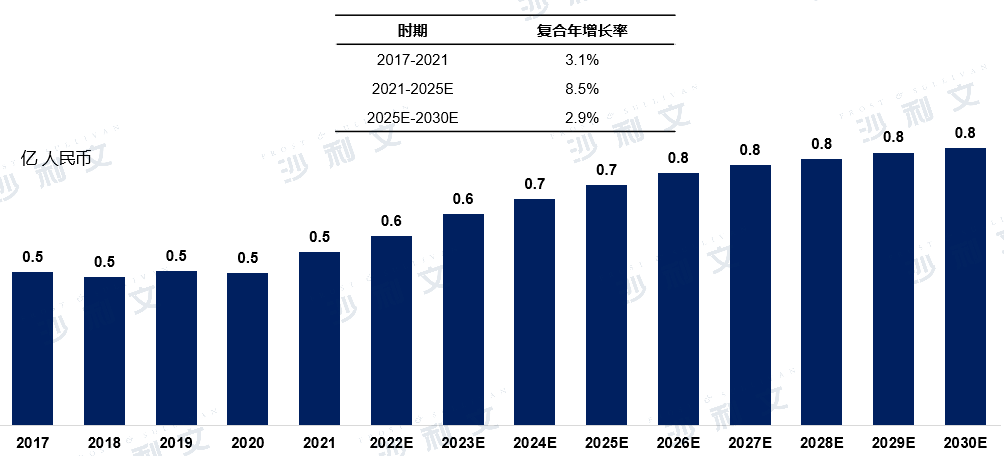
Data source: Analysis by Frost & Sullivan
Market Overview of Patent Ductus Arteriosus Occluders
In 2021, the market size of transcatheter occluders for patent ductus arteriosus in China reached 58.9 million RMB, with a compound annual growth rate of 0.4% from 2017 to 2021. It is estimated that by 2025, this figure will reach 88.4 million RMB, with a compound annual growth rate of 10.3% from 2021 to 2025. By 2030, the market size is expected to reach 107.3 billion RMB.
The market scale and forecast of transcatheter occluders for patent ductus arteriosus in China
2017 to 2030 (estimated)
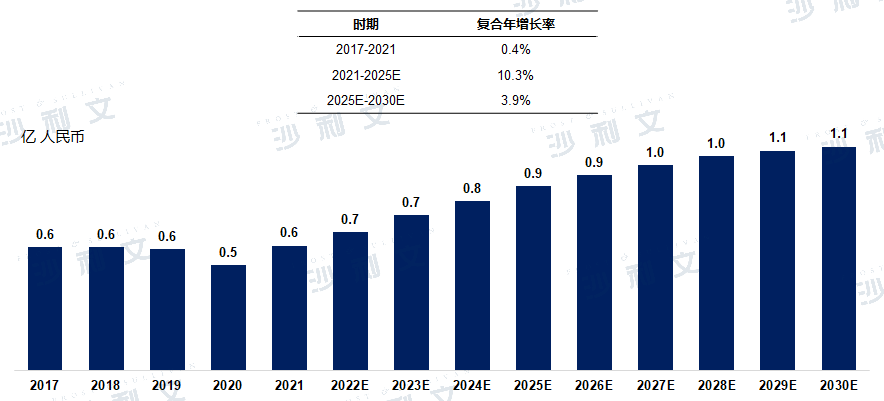
Data source: Analysis by Frost & Sullivan
Epidemiology of Cardiogenic Stroke in China
Due to the aging population in China, the number of cases of cardioembolic stroke in China increased from 3.9 million in 2017 to 4.5 million in 2021. It is estimated that from 2025 to 2030, the number will grow from 5 million to 5.7 million, with a compound annual growth rate of 2.5%. Stroke related to atrial fibrillation (with or without other cardiovascular diseases) accounts for 79% of all heart strokes.
Epidemiology of Cardiogenic Stroke in China2017 to 2030 (estimated)
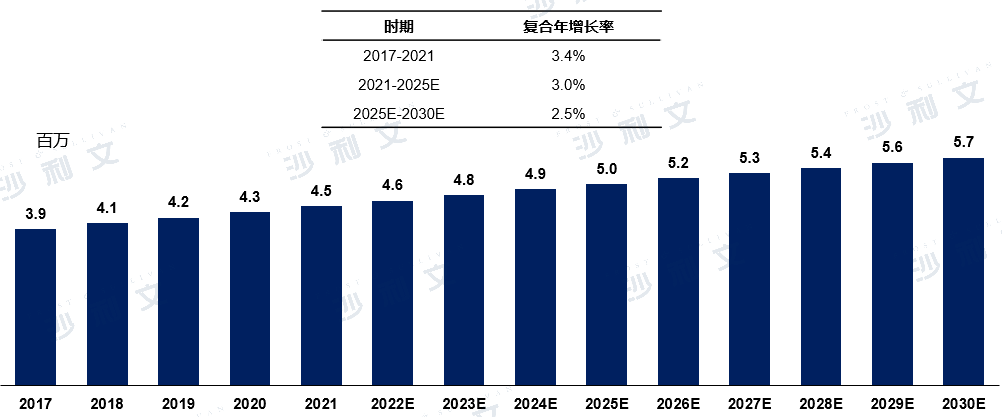
Data source: Analysis by Frost & Sullivan
Overview of the Medical Device Market for Left Atrial Appendage Occluders in China
The market for left atrial appendage occluders has maintained a rapid growth trend from 2017 to 2030. In China, the market size for left atrial appendage occluders increased from 600 million yuan in 2017 to 5000 million yuan in 2021, with a compound annual growth rate of 70.8%. The market size will continue to rise, expected to reach 1.6 billion yuan by 2025, with a compound annual growth rate of 35.0% from 2025 to 2030.
Overview of the Medical Device Market for Left Atrial Appendage Occluders in China
2017 to 2030 (estimated)
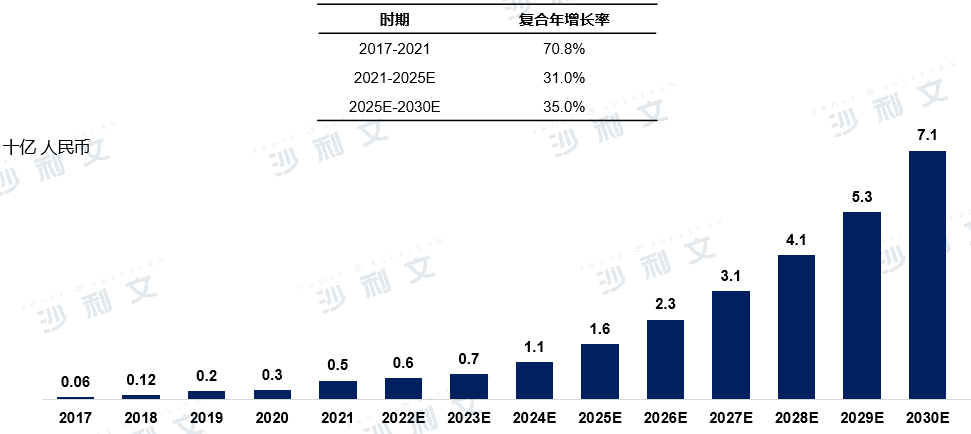
Data source: Analysis by Frost & Sullivan
Overview of the Medical Device Market for Patent Ductus Openings Closure Devices in China
Similar to the left atrial appendage occluder, the market for the Chinese oval foramen occluder has been growing rapidly. The market size of the oval foramen occluder increased from 18.9 million yuan in 2017 to 190 million yuan in 2021, with a compound annual growth rate of 77.3%. The market size will continue to rise, expected to reach 370 million yuan by 2025 and 760 million yuan by 2030, with a compound annual growth rate of 15.5% from 2025 to 2030.
Overview of the Medical Device Market for Patent Ductus Openings Closure Devices in China
2017 to 2030 (estimated)
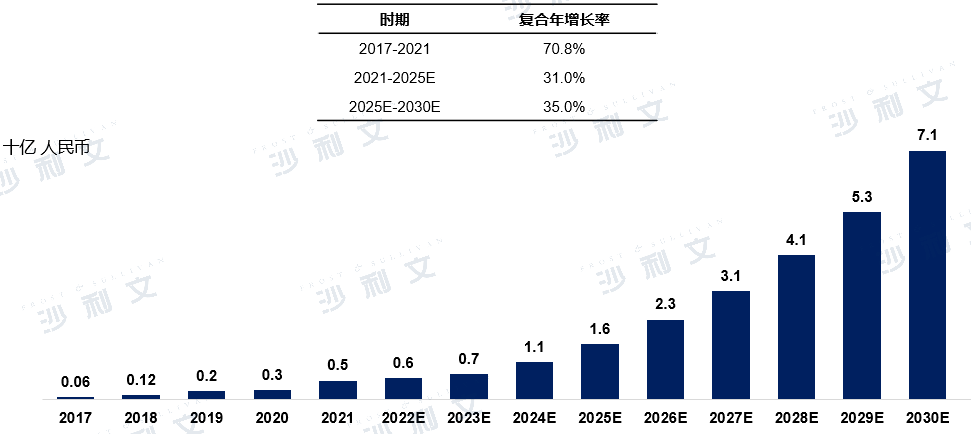
Data source: Analysis by Frost & Sullivan
Frost & Sullivan, integrating 61 years of global consulting experience, has been serving the booming Chinese market with dedication for 24 years. With a global perspective, it demonstrates how clients accelerate their corporate growth and achieve benchmark positions in industry growth, innovation, and leadership. The healthcare industry is one of Frost & Sullivan's core areas of focus. Over the past 20-plus years, the Frost & Sullivan team has provided financing financial advisory, IPO industry advisory, strategic consulting, management consulting, and other services to hundreds of outstanding domestic and international biopharmaceuticals, medical devices, healthcare services, and internet healthcare companies. Successful listings include: Jian Shi Technology (9877.HK), Health Yuan (JCARE.SW), Lepu Medical (LEPU.SW), Ding Dang Health (9886.HK), BAIOSYTHU (2315.HK), Zhiyun Health (9955.HK), MeinGene (6667.HK), Prenetics (PRE.NASDAQ), Yunkang Group (2325.HK), Ruike Biotech (2179.HK), Lepu Biotech (2157.HK), Clear Medical (1406.HK), BaiXinAn (2185.HK), Yonghe Medical (2279.HK), KailaiYing (6821.HK), Beihai Kangcheng (1228.HK), Gusheng Tang (2273.HK), YingTong Technology (2251.HK), Clover Biotech (2197.HK), Minimally Invasive Robotics (2252.HK), Harmony Cayman (2256.HK), Kunbo Medical (2216.HK), Xianruida (6669.HK), Kangsheng Global (9960.HK), Yimaitong (2192.HK), Tengsheng Bovy (2137.HK), CanSino (2162.HK), Chaopu Ophthalmology (2219.HK), Guichuang Tongqiao (2190.HK), Hua Huang Medicine (0013.HK), Koi Pharmaceutical (2171.HK), Zhaoke Ophthalmology (6622.HK), Nature Pharmacy (UPC.NASDAQ), Saiseng Pharmaceutical (6600.HK), Zhaoyan New Drug (6127.HK), Novogene Health (6606.HK), Tianyan Pharmaceutical (ADAG.NASDAQ), Beikang Medical (2170.HK), JianBimiaoMiao (2161.HK), Minimally Invasive HeartLink (2160.HK), RuiLi Medical Beauty (2135.HK), Gaoke Pharmaceutical (1167.HK), Hepo Pharma (2142.HK), JD Health (6618.HK), Deqi Pharmaceutical (6996.HK), Rongchang Biotech (9995.HK), WuXi AppTec (2126.HK), XiangSheng Pharmaceutical (2096.HK), Yunding New Energy (1952.HK), Jiahe Biotech (6998.HK), ZaiDing Pharmaceuticals (9688.HK), Ocular Biotechnology (1477.HK), Yongtai Biotech (6978.HK), Haipure Pharmaceutical (9989.HK), Kuaibo Pharmaceuticals (9939.HK), Peijia Medical (9996.HK), Kangfang Biotech (9926.HK), NuoCheng Jianhua (9969.HK), Tianjing Biotech (IMAB.NASDAQ), Kanglong Chemical (3759.HK), China Antibody (3681.HK), Dongyao Pharmaceutical (1875.HK), Yasheng Pharmaceutical (6855.HK), Fuhong Hanlin (2696.HK), Hansoh Pharmaceutical (3692.HK), Mabtech Pharmaceuticals (2181.HK), Fangda Holdings (1521.HK), Via Bio (1873.HK), CStone Pharmaceuticals (2616.HK), Junshi Biosciences (1877.HK), WuXi AppTec (2359.HK), Innovent Biologics (1801.HK), Huading Medicine (2552.HK), BeiGene (6160.HK), Gilead Sciences (1672.HK), WuXi Biologics (2269.HK), China Resources Medicine (3320.HK), Yakuten Scientific & Pharmaceutical (2633.HK), Hua Huang China Medicine (HCM.NASDAQ), Biotechnology (1548.HK), BBI Life Sciences (1035.HK), etc. In terms of the number of filings, the Frost & Sullivan healthcare team maintains an absolute leading position in Hong Kong's healthcare IPO market, consistently ranking first in market share from 2018 to 2021.
Since the listing of the first batch of companies on the Sci-tech Innovation Board in July 2019, Frost & Sullivan reports have been widely cited in the prospectuses of leading Sci-tech Innovation Board listed companies in the industry. These include: Kangwe Century (688426.SH), Jinqian Protein (688137.SH), Nuo Cheng Jianhua (688428.SH), Aopu Mai Biotech (688293.SH), MicroPort Electrodiagnostics (688351.SH), Mengke Pharmaceutical (688373.SH), Yifang Biotech (688382.SH), Jicui Yaokang (688046.SH), Haichuang Pharmaceutical (688302.SH), Rongchang Biotech (688331.SH), Rendu Biotech (688193.SH), Shouyao Holdings (688197.SH), Heyuan Biotech (688238.SH), Yaxin Security (688225.SH), Xidi Micro (688173.SH), Mawei Biotech (688062.SH), Yahong Medicine (688176.SH), BeiGene (688235.SH), Jiahe Meikang (688246.SH), Dizhe Medicine (688192.SH), Novogene (688105.SH), Chengda Biotech (688739.SH), Geke Micro (688728.SH), Huaxi Biotech (688363.SH), Junshi Biotech (688180.SH), Zeping Pharmaceutical (688266.SH), BeiGene (688177.SH), Shenzhou Cells (688520.SH), and others. They are considered to be one of the most powerful, professional, and influential industry research institutions in the sector. We hope to work with enterprises to understand industry trends, seize development opportunities, jointly promote innovation and upgrading of China's health industry, and build a healthy future.
Recommended Reading
08. Frost & Sullivan assists Prenetics in successfully going public in the US (NASDAQ: PRE)
32. Frost & Sullivan assists Nature's Way Pharmaceuticals in successfully going public on the NASDAQ:UPC)
58. Frost & Sullivan assists Tianjing Biology in successfully going public in the US (NASDAQ: IMAB)

