Nuo Cheng Jianhua Medicine Co., Ltd. (Stock Code:688428.SH) on2022year9month21The company successfully logged into the Sci-Tech Innovation Board on the same day. It is an innovative drug enterprise focusing on areas such as oncology and autoimmune diseases, and has currently established product lines targeting multiple popular targets. Frost & SullivanFrost & SullivanFrost & Sullivan, hereinafter referred to as 'Frost & Sullivan', hereby warmly congratulates the company on its successful listing.

Nuo Cheng Jianhua Medicine Co., Ltd. (hereinafter referred to as 'Nuo Cheng Jianhua Medicine') was established on2022year9month21The company was successfully listed on the stock market, with the number of shares issued being176,43210,000 shares, issue price per share11.03yuan.
The company is an innovative pharmaceutical enterprise focusing on areas such as oncology and autoimmune diseases, and has currently established a product pipeline targeting multiple popular targets. As an innovation-driven company, the company takes it as its responsibility to improve the quality of life and health levels for all humanity. Guided by unmet clinical needs in the global pharmaceutical market, the company aims to develop the most breakthrough drugs among peers or first-to-market products. Under the leadership of a management team with rich experience in R&D, production, and commercialization, an integrated biopharmaceutical platform has been constructed. The platform balances R&D quality with speed, establishing a product pipeline that is highly innovative and risk-averse, covering multiple highly promising hot targets. Frost & Sullivan has long been monitoring the global and Chinese biopharmaceutical industries, publishing a large number of research reports, which are widely cited in the prospectuses of leading science and technology innovation board-listed companies in the industry, helping clients accelerate growth.
Antitumor Drug Market Overview
In the Chinese pharmaceutical market, the sales of anti-tumor drugs have been showing a steady growth trend in recent years.2016Year-end2020In the year, the market scale was2020RMB in year1,975yuan, in the past5The compound annual growth rate for the year reached12.1%The progress in cancer treatment methods has also led to an upward trend in the Chinese anti-tumor drug market over the next few years. It is expected that the Chinese anti-tumor drug market2025The annual amount will reach RMB4,1621000 million yuan, with an annual compound growth rate of16.1%, to2030Annual achievement6,831100 million yuan.
Market Scale and Forecast of Antitumor Drugs in China
2016Year to2030Year (estimated)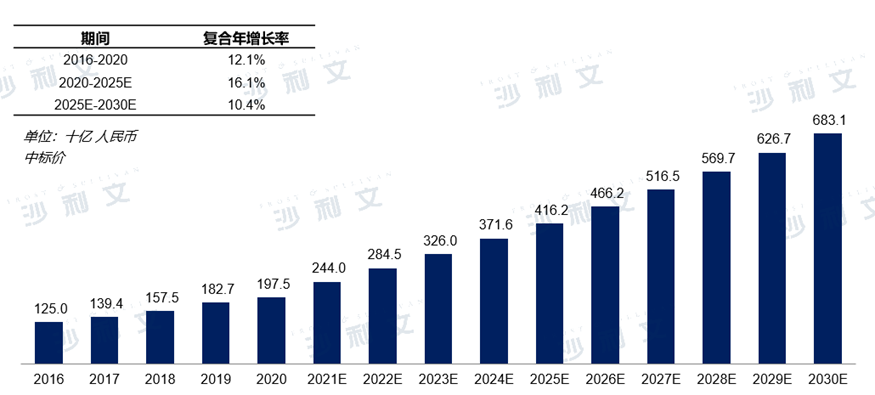
Data source: Analysis by Frost & Sullivan
Autoimmune disease drug market
Autoimmune diseases are a type of illness where the body's immune system mistakenly attacks itself, possibly due to an abnormal low or high activity of the immune system. Approximately100Different types of autoimmune diseases can affect almost any part of the body, including the heart, brain, nerves, muscles, skin, eyes, joints, lungs, kidneys, glands, digestive tract, and blood vessels.
Common autoimmune diseases include rheumatoid arthritis, ankylosing spondylitis, psoriasis, lupus erythematosus, etc. These diseases usually have a recurrent course and are characterized by a chronic protracted process. The main drugs for treating autoimmune system diseases include non-steroidal anti-inflammatory drugs, glucocorticoids, and immunosuppressants. Currently, there is no cure for autoimmune diseases.
With a deeper understanding of the pathophysiology and related biological pathways of autoimmune diseases, there is hope that more innovative biologics and small molecule targeted drugs will be developed in the future to offer patients with autoimmune diseases more treatment options, such as antiIL-6antibody,BTKInhibitors, etc. These newly developed drugs not only provide more available medications for patients with autoimmune diseases such as rheumatoid arthritis and systemic lupus erythematosus, but also help address more therapeutic areas.
China has a large number of patients with autoimmune diseases, and it is reported that the number is on the rise.2020In [year], the number of patients with systemic lupus erythematosus, ulcerative colitis, and rheumatoid arthritis in China was approximately104ten thousand,43Wanhe596Tens of thousands. With the development and improvement of diagnostic technologies for autoimmune diseases in our country, the market demand for healthcare services will be stimulated in the coming years. It is expected2025The overall market scale for the year will reach87billion US dollars2020Year-end2025The annual compound annual growth rate for28.1%.
Market Scale and Forecast of Drugs for Autoimmune Diseases in China
2016Year-end2030Year (estimated)

Data source: Analysis by Frost & Sullivan
BTKInhibitor Market Overview
BTKIt is a cytoplasmic non-receptor tyrosine kinase,BImmune cells such as cells, macrophages, and microglia play an important role in the development and physiological functions of immune cells involved in the pathological process of autoimmune diseases.BAfter cellular lymphoma becomes cancerous,BTKIt will become extremely active,BTKActivation of the signaling pathway sends out a startBSignaling for the growth of malignant tumor cells in cells, andBCells play an important role in the growth, migration, and other processes of malignant tumors.
BTKInhibitors act byBCRsignal pathway, andBTKCombined inhibitionBTKself-phosphorylation, inhibitionBTKActivation thereof can block cell conduction and induce apoptosis, inhibit the growth of malignant tumors, and achieveBThe regulatory role in the development of cellular tumors has become a popular target for treating hematological malignancies and autoimmune disorders.
Currently, ChinaBTKThe inhibitor market mainly consists of three drugs: Imbrutinib (Janssen), Zanubrutinib (BeiGene), and Osimertinib (NuoChegenghua). Since the firstBTKinhibitor2017After its approval in China last year, ChinaBTKThe market is growing rapidly, and so2020Year-on-year growth to13100 billion RMB, and it is expected to58.6%The compound annual growth rate is2025Year-on-year growth to131100 billion RMB, this market will be2030expanded to2251000 million RMB2025till2030The compound annual growth rate for the year is11.5%.
chinaBTKMarket size and forecast of inhibitors
2017Year to2030Year (estimated)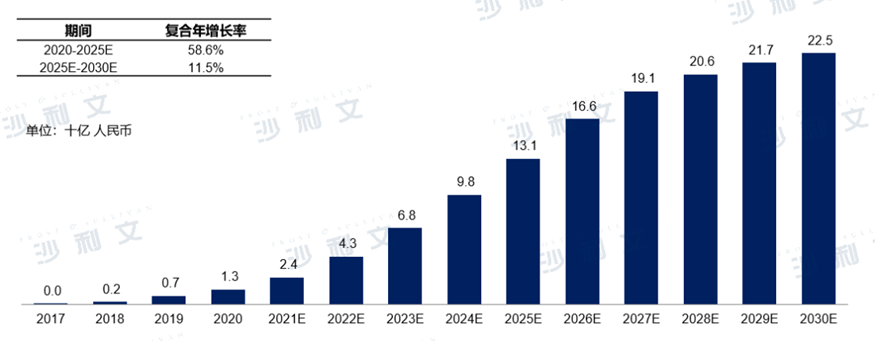
Data source: Analysis by Frost & Sullivan
FGFRInhibitor Market Overview
fibroblast growth factor receptor(FGFRs)It is a highly conserved, widely distributed transmembrane tyrosine kinase receptor containing four subtypesFGFR-1,2,3and4).FGFRSignal dysregulation can lead to the occurrence, development, proliferation, survival, and metastasis of cancer. Almost every type of malignant tumor hasFGFRdistortion(Amplification, mutation, rearrangement),7.1%exists in solid tumorsFGFRDeformity. Most commonly seen in urothelial carcinoma(31.7%)Cholangiocarcinoma(25.2%)hepatocellular carcinoma(30.0%)breast cancer(17.5%)and gastric cancer(6.7%).
FGFRInhibitors block intracellular kinases withATPCombined with its activity, it blocks downstream signaling pathways, thereby exerting anti-tumor effects such as anti-cell proliferation and anti-angiogenesis.FGFRAbnormal signal pathways have become an important cause of various tumors and show promise as new targets. At the same time,FGFRActivation of signaling pathways can also lead to the development of resistance in some tumor cells to targeted therapies. ThereforeFGFRIt has become a hot area in the research and development of tumor-targeting drugs.
Currently, there are threeFGFRThe inhibitor has been approved for two different indications. ErdavitinibBalversa(Erdafitinib) has beenFDAPemitinib and imfiniratin are approved for the treatment of metastatic urothelial carcinoma, while pembrolizumab is approved for the treatment of cholangiocarcinoma.
FGFTMechanism of action of inhibitors
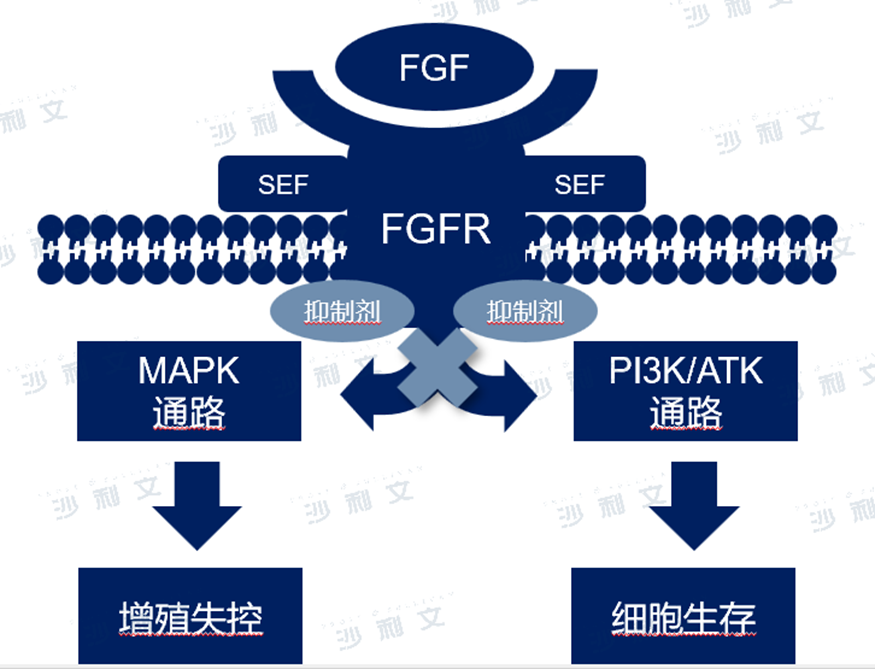
Data source: Literature review and analysis by Frost & Sullivan
TRKInhibitor Market Overview
TRKKinases are considered to be involved in the growth, differentiation, and apoptosis of neuronal cells. Under normal circumstances, allTRKProteins all have a similar extracellular domain structure, but each has different ligands. These proteins are usually expressed in the nervous system and are activated upon signal induction.TRKPhosphorylation occurs and activates downstream signaling pathways. WhenTRKWhen dysregulation leads to the overactivation of downstream pathways, it may contribute to cancer development.TRKKinases in melanoma, non-small cell lung cancerNSCLCthyroid cancer, malignant myeloid leukemiaAMLOverexpression has been observed in various types of cancer, including glioblastoma, astrocytoma, colon cancer, etc., and this overexpression is closely related to the migration of tumor cells.
TRKThere are mainly three types of inhibitors. One is the tyrosine kinase inhibitor, which can inhibit a variety of kinases.TKIDrugs, with a large number of marketed products, including entrectinib, crizotinib, latratinib, ponatinib,MGCD516,TSR-011Among them, entrectinib holds a leading position. One is for specific inhibitionTRKThe representative drug is larotinib. There is another possibility that it may targetTRKFused non-specificityTKIsIncluding cabozantinib and regorafenib.
TRKThe market scale of inhibitors in China is2022Annual target0.1billions, expected to2025Year will be180.4%The compound annual growth rate reached2.1billion dollars, to2030Year will be39.9The compound annual growth rate increased to11.4billion dollars.
chinaTRKMarket size and forecast of inhibitors
2016Year to2030Year (estimated)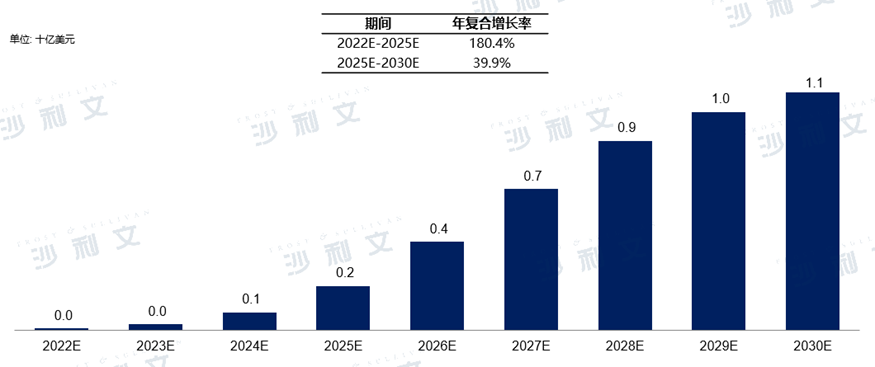
Data source: Analysis by Frost & Sullivan
CD19Overview of the Targeted Drug Market
CD19throughBCell receptorB cell receptor, BCR) Adjustment by dependency and non-dependencyBCell development, proliferation, and differentiation. Currently approvedCD19The indications for targeted therapies are mainly focused onDLBCLandB-ALL.CD19As normal and malignantBLymphocyte-specific surface markers, which are expressed in a limited manner in normal tissues andBThe widespread expression of lymphoma cells reduces off-target toxicity to a certain extent, making itBA highly promising target molecule for immunotherapy of lymphocyte malignancies.
chinaBThe number of newly diagnosed cases of malignant tumors in cells is continuously increasing. In China,CD19Approved IndicationsDLBCLThe number of patients is in2020yearly21.1Tens of thousands, expected in2025year and2030reached in25.9Ten Thousand People and29.9ten thousand people. At the same time,CD19The indications for targeted therapies are also moving towardsBThe scope of cellular malignancies is constantly expanding beyond that. This will further expand.CD19The patient pool for targeted drugs. The growing patient base will increase market demand forCD19The demand for targeted drugs, thereby drivingCD19The development of the targeted drug market.
![]()
Frost & Sullivan integrates the globe.61years of consulting experience,24Frost & Sullivan has been dedicated to serving the booming Chinese market for years, demonstrating a global perspective in helping clients accelerate their business growth and achieve industry-leading benchmarks in terms of growth, innovation, and leadership. The health industry is one of the core areas of focus for Frost & Sullivan. Recently20For many years, the Frost & Sullivan team has provided financing and financial advisory services to hundreds of outstanding domestic and international biopharmaceutical, medical device, healthcare services, and internet healthcare companies.IPO listingIndustry consulting, strategic consulting, management consulting, and other services. Successful listing cases include: Dingdang Health (9886.HK), Baidu Map Suggest (2315.HK), Zhiyun Health9955.HK), MeinGene6667.HK),Prenetics(PRE.NASDAQ), Yunkang Group2325.HK), Ruike Biotechnology2179.HK), Leepu Biotech2157.HK), Clear Medical (1406.HK), Baisinkan2185.HK), Yonghe Medical (2279.HK), Kailaiying6821.HK), Beihai Kangcheng1228.HK), Gusheng Hall2273.HK), Eagle Eye Technology2251.HK), Clover Biotechnology2197.HK), minimally invasive robots2252.HK), and Yuhuan Cayman2256.HK), Kunbo Medical (2216.HK), Xianruida6669.HK), Kangsheng Global9960.HK), Medical Pulse Connect (2192.HK), Tengsheng Bo Pharmaceutical2137.HK), Conoco (2162.HK), Chaogju Eye Hospital (2219.HK), Guichuangtong Bridge2190.HK), and Huang Medicine0013.HK), Keji Pharmaceutical2171.HK), Zhaoke Eye Hospital6622.HK), Nature MedicineUPC.NASDAQ), SinoBiologics (6600.HK), Zhaoyan New Drugs6127.HK), Novo Nordisk Health (6606.HK), Tianyan PharmaceuticalADAG.NASDAQ), Beikang Medical (2170.HK), Jianbimiao Miao Miao2161.HK), Minimally Invasive Cardiac Angioplasty (2160.HK), Ruili Medical Beauty2135.HK), Jiake Pharmaceutical1167.HK), and Bopan Medicine2142.HK), JD Health6618.HK), Deqi Medicine6996.HK), Rongchang Biology9995.HK), WuXi AppTec Genomics (2126.HK), Sinobiopharmaceuticals2096.HK), Yunding Xinyao1952.HK), Harvest Biotech6998.HK), ZaiDi Pharma9688.HK), Ocumivir1477.HK), Yongtai Biology6978.HK), Hype Pharma9989.HK), Kechuang Pharmaceutical9939.HK), Peijia Medical (9996.HK), Kangfang Biology9926.HK), Novartis HealthCare (9969.HK), Celestial BeingsIMAB.NASDAQ), Kanglonghua Cheng3759.HK), China Antibody3681.HK), Dongyao Pharmaceutical1875.HK), Yasheng Medicine6855.HK), Fosun Pharma (2696.HK), Hansoh Pharmaceutical3692.HK), Mabotech2181.HK), Founder Holdings1521.HK), Viva Biology1873.HK), CStone Pharmaceuticals2616.HK), Junshi Biology1877.HK), WuXi AppTec (2359.HK), Innovent Biologics1801.HK), Hualing Medicine2552.HK), BeiGene (6160.HK), Gelifarmaceuticals1672.HK), WuXi AppTec Biologics2269.HK), China Resources Medicine3320.HK), Yakult Biopharmaceuticals2633.HK), and Huang China MedicineHCM.NASDAQ), Biotechnology1548.HK),BBILife Sciences1035.HK) and so on. In terms of the number of submissions, Frost & Sullivan's healthcare team in Hong Kong is involved inIPO listingMaintain an absolute leading position,2018till2021Year after year, it has always ranked first in market share.
since2019year7Since the initial listing of the first batch of companies on the Sci-tech Innovation Board, Frost & Sullivan reports have also been widely cited in the prospectuses of leading Sci-tech Innovation Board-listed companies in the industry, including: OPPOMABIO (688293.SH), minimally invasive electrophysiology688351.SH), Mengke Pharmaceutical688373.SH), Yifang Biology688382.SH), Jicui Pharmaceutical Health (688046.SH), Haiscot Biopharmaceuticals688302.SH), Rongchang Biology688331.SH), Rendu Biology688193.SH), Capital Pharmaceutical Holdings (688197.SH), and Yuanbiotech (688238.SH), YASIN Security688225.SH), Xidiwei688173.SH), MyWay Biotechnology688062.SH), Yarong Medicine688176.SH), BeiGene (688235.SH), Jiahemeikang688246.SH), DiZhe Medicine688192.SH), Novozymes688105.SH), Chengda Biology688739.SH), Gecko Micro688728.SH), Huaxi Biology688363.SH), Junshi Biology688180.SH), Zhejiang Genomics & Therapeutics Co., Ltd.688266.SH), Bai'ao Tai688177.SH), SinoCell (688520.SH) and other institutions are considered one of the most powerful, professional, and influential industry research organizations in the sector. We aim to work with enterprises to understand industry trends, seize development opportunities, jointly promote innovation and upgrading of China's big health industry, and build a healthy future.
Recommended Reading
01.Frost & Sullivan assisted Dingdang Health in successfully going public in Hong Kong(9886.HK)
03.Frost & Sullivan assists Zhiyun Health in successfully going public in Hong Kong(9955.HK)
05.Frost & Sullivan assistancePreneticsSuccessfully listed in the US(NASDAQ:PRE)
06.Frost & Sullivan assisted YunKang Group in successfully going public in Hong Kong(2325.HK)
09.Frost & Sullivan helps Clear Medical successfully go public in Hong Kong(1406.HK)
15.Frost & Sullivan assisted Yingpeng Technology in successfully going public in Hong Kong(2251.HK)
18.Frost & Sullivan assisted Yuhuan in successfully listing on the Hong Kong Stock Exchange(2256.HK)
19.Frost & Sullivan assisted Kunbo Medical in successfully going public in Hong Kong(2216.HK)
20. Frost & Sullivan assisted Xianruida in successfully going public in Hong Kong(6669.HK)
21.Frost & Sullivan assisted Kangsheng Global in successfully going public in Hong Kong(9960.HK)
23.Frost & Sullivan assisted ConocoAL on its successful listing in Hong Kong(2162.HK)
24.Frost & Sullivan assisted Chaoyu Eye Hospital in successfully going public in Hong Kong(2219.HK)
25.Frost & Sullivan assisted Guichuangtongqiao in successfully going public in Hong Kong(2190.HK)
29.Frost & Sullivan assisted Nature Pharma in successfully going public in the US(NASDAQ:UPC)
34.Frost & Sullivan assisted Jianbimiao Miao in successfully going public in Hong Kong(2161.HK)
36.Frost & Sullivan assisted RuiLi Medical Beauty in successfully going public in Hong Kong(2135.HK)
45.Frost & Sullivan assisted Jiahé Biotech in successfully going public in Hong Kong(6998.HK)
51.Frost & Sullivan assisted Peijia Medical in successfully going public in Hong Kong(9996.HK)
52.Frost & Sullivan assisted Kangfang Biotech in successfully going public in Hong Kong(9926.HK)
54.Frost & Sullivan assisted Tianjing Biology in successfully going public in the US(NASDAQ: IMAB)
59.Frost & Sullivan assisted Fuhong Hanlin in successfully going public in Hong Kong(2696.HK)
63.Frost & Sullivan assisted ViaBio in successfully going public in Hong Kong(1873.HK)
66.Frost & Sullivan assisted WuXi AppTec in successfully going public in Hong Kong(2359.HK)

