Shanghai Mengke Pharmaceutical Co., Ltd. (Stock Code:688373.SH) on2022year8month5The company successfully listed on the Sci-tech Innovation Board. It is an innovative pharmaceutical enterprise with a focus on treating infectious diseases, possessing global independent intellectual property rights and international competitiveness. Frost & SullivanFrost & Sullivan,Frost & Sullivan (hereinafter referred to as 'Frost & Sullivan') hereby warmly congratulates the company on its successful listing.

Shanghai Mengke Pharmaceutical Co., Ltd. (hereinafter referred to as 'Mengke Pharmaceutical') was established on2022year8month5The company was successfully listed on the stock market, with the number of shares issued being13,000.0010,000 shares, issue price per share8.16yuan.
Mengke Pharmaceutical is an innovative drug company centered on the treatment of infectious diseases, with global independent intellectual property rights and international competitiveness. It is committed to discovering, developing, and commercializing innovative drugs for unmet clinical needs. The company has established R&D centers in China and the United States and possesses an international core R&D team. The company's R&D team has many years of experience in international innovative drug research and management, having led or participated in the development of several marketed new anti-infective drugs. The company adheres to independent research and development, delves into specialized niche areas, and has established an integrated antibacterial new drug R&D system within the company, covering all aspects of new drug development including early design and screening, preclinical evaluation, global clinical development, registration application, and production management.
The company's first antibacterial drug product, Contizolam Tablets (trade name: Uxetia), is a new generation oxazolidone antibacterial drug designed and developed independently by the company. It can be used to treat infections caused by multidrug-resistant Gram-positive bacteria. Compared with existing drugs, Contizolam not only ensures excellent efficacy against multidrug-resistant Gram-positive bacteria but also improves drug safety. It provides doctors and patients with a safer medication choice and better treatment plans. Contizolam has the potential to replace current similar antibacterial drugs and is expected to become the clinical first choice for treating multidrug-resistant Gram-positive bacteria in the future. Contizolam has been2021year6month1Approved for marketing through the priority review and approval process of the National Medical Products Administration on2022year1Monthly medical insurance enrollment. In addition, the company has multiple projects that have entered the clinical phase and/Or core products in the commercialization phase and new anti-drug-resistant bacterial drugs in preclinical trials, as well as pipelines for kidney cancer, nephritis drugs, and anti-COVID-19 drugs.
Frost & Sullivan has long been focusing on the global and Chinese biopharmaceutical industries, publishing a large number of research reports that are widely cited in the prospectuses of leading science and technology innovation board listed companies in the industry, helping clients accelerate growth.
Analysis of the Antibacterial Drug Market in China
2016Year to2019In [year], the market scale of antibacterial drugs in China showed a stable growth trend.2020Affected by the pandemic last year, sales of antibacterial drugs were impacted due to a reduction in bacterial infectious diseases caused by wearing masks. As a result, the antibacterial drug market in China2019year1,662RMB 10 billion decreased to1,224100 million RMB. From2016Year-end2020In [year], the market scale of antibacterial drugs in China from RMB1,535RMB 100 million decreased to RMB1,244yuan, with a compound annual growth rate of-5.1%. Forecast to2025In [year], the market will reach RMB1,380yuan, from2020Year-end2025Annual Compound Annual Growth Rate2.1%.
Market scale of antibacterial drugs in China2016-2030E
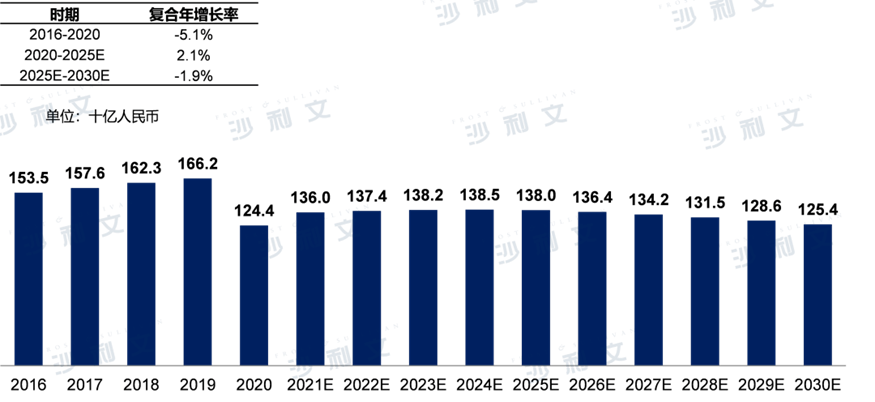
Data source: Analysis by Frost & Sullivan
Multidrug-resistant Gram-positive bacteria in China
infected Antibacterial drug market analysis
Antibiotics for infections caused by multidrug-resistant Gram-positive bacteria are mainly empiric in use in China. In the future, the number of days of antibiotic treatment for multidrug-resistant Gram-positive bacterial infections in China is expected to continue to increase.2025Year-on-year growth to1,610Ten thousand days,2020Year to2025The compound annual growth rate for the year is7.4%,2030The number will increase in the new year2,140Ten thousand days,2025Year-end2030The compound annual growth rate for the year is5.9%.
Treatment duration of antibacterial drugs for the treatment of multidrug-resistant Gram-positive bacterial infections in China2016 - 2030E
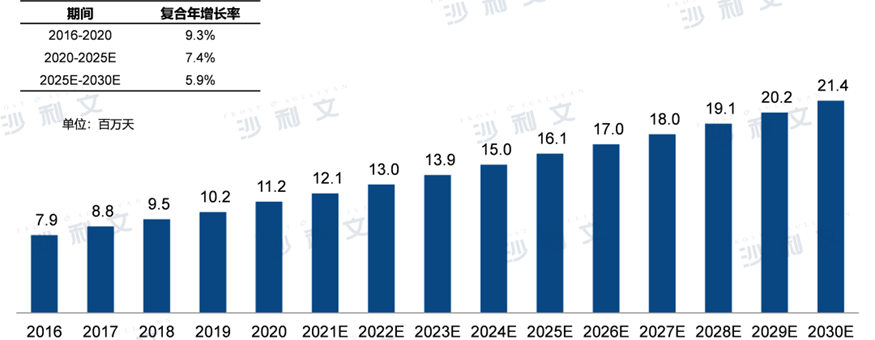
Data source: Analysis by Frost & Sullivan
China is one of the largest markets for antibacterial drugs against multidrug-resistant Gram-positive bacterial infections globally.2020The annual market scale is41RMB 10 billion,2016Year-end2020The compound annual growth rate for the year is6.1%Although compared to2019There was a slight decline in the year, mainly due to the outbreak of the pandemic. The widespread use of masks has reduced the number of people infected with bacteria. However, driven by favorable government policies and continuously increasing R&D expenses, the market scale of antibacterial drugs in China will be2021It started to show an increasing trend at the beginning of2025Year-on-year increase82RMB 10 billion2030Year-on-year increase108RMB 10 billion,2025Year-end2030The compound annual growth rate for the year is5.8%.
Azoleketone drugs account for a certain proportion of the antibacterial drug market for multidrug-resistant Gram-positive bacterial infections in China, and there is a growth trend in this area.2020The market scale of azoleketone antibacterial drugs in China in15RMB 10 billion, higher than2016year7RMB 10 billion, with a compound annual growth rate of21.2%The market scale of azoleket antibacterial drugs in China will be2025Year-on-year increase39RMB 10 billion,2030Year-on-year increase60RMB 10 billion2025Year to2030The compound annual growth rate for the year is8.7%.
The market for antibacterial drugs in China for treating multidrug-resistant Gram-positive bacterial infections 2016 - 2030E
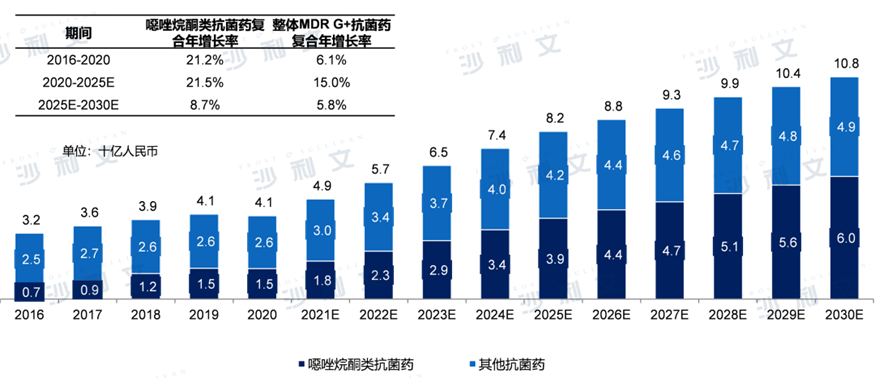
Data source: Analysis by Frost & Sullivan
Multidrug-resistant Gram-negative bacteria
Analysis of the Antibacterial Drug Market for Infections
The total treatment days of antibacterial drugs for multidrug-resistant Gram-negative bacterial infections in China decreased from2016year3,360Ten thousand days to grow to2020year4,690Ten thousand days, with the compound annual growth rate during this period8.8%The number of days of antibacterial drug treatment for multidrug-resistant Gram-negative bacterial infections in China is expected to continue increasing in the future.2025Year-on-year growth to8,730Ten thousand days,2020Year-end2025The compound annual growth rate for the year is13.2%,2030The number will increase in the new year1.1100 million days,2025Year-end2030The compound annual growth rate for the year is5.2%.
Treatment duration of antibacterial drugs for treating multidrug-resistant Gram-negative bacterial infections in China2016-2030E
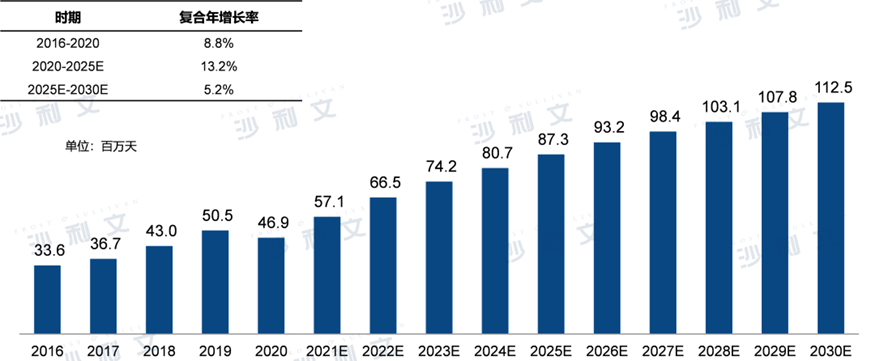
Data source: Analysis by Frost & Sullivan
2016 - 2019In [year], the market for antibacterial drugs against multidrug-resistant Gram-negative bacterial infections in China maintained stable growth.2020Due to the pandemic, the market for antibacterial drugs for multi-drug resistant Gram-negative bacterial infections in China decreased slightly compared to the previous year, but overall it still showed an upward trend.2016year140RMB 10 billion increased2020year189RMB 10 billion, with a compound annual growth rate of7.9%. From2020Year-end2025In the year, the market will grow to339RMB 10 billion, with a compound annual growth rate of12.4%, to2030The final year will reach422RMB 10 billion.
The market for antibacterial drugs in China for treating multidrug-resistant Gram-negative bacterial infections2016-2030E
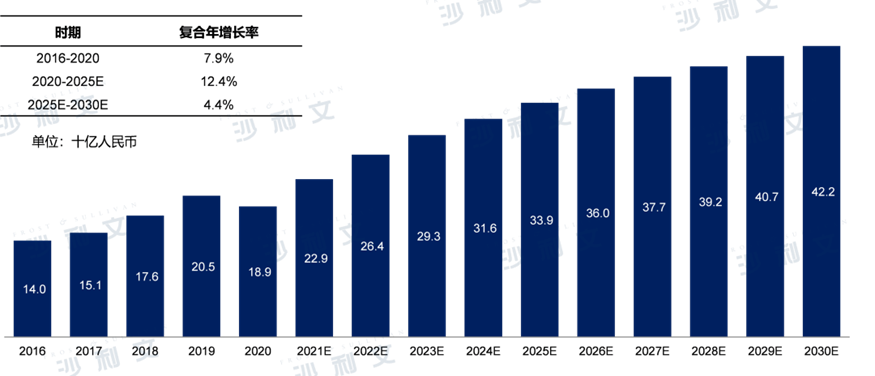
Note: Gram-negativeMDRAntibiotics include carbapenems, β-Lactamase/β-Lactamase inhibitor combinationBL/BLI), tetracycline and polymyxin antibacterial drugs.
Data source: Analysis by Frost & Sullivan
Frost & Sullivan integrates globally.61years of consulting experience,24Frost & Sullivan is dedicated to serving the booming Chinese market with a global perspective, helping clients accelerate their business growth and achieve benchmark positions in industry growth, innovation, and leadership. The Greater Health Industry is one of the core areas of focus for Frost & Sullivan's research. Recently20For many years, the Frost & Sullivan team has provided financing and financial advisory services to hundreds of outstanding domestic and international biopharmaceutical, medical device, healthcare services, and internet healthcare companies.IPO listingIndustry consulting, strategic consulting, management consulting, and other services. Successful listings include: Zhiyun Health (9955.HK), MeinGene6667.HK),Prenetics(NASDAQ: PRE), Yunkang Group2325.HK), Ruike Biotechnology2179.HK), Leepu Biotech2157.HK), Clear Medical1406.HK), Baisinkan2185.HK), Yonghe Medical (2279.HK), Kailaiying6821.HK), Beihai Kangcheng1228.HK), Gusheng Hall2273.HK), Eagle Eye Technology2251.HK), Clover Biotechnology2197.HK), minimally invasive robots2252.HK), and Yuhuan Cayman2256.HK), Kunbo Medical (2216.HK), Xianruida6669.HK), Kangsheng Global9960.HK), Medical Pulse Connect (2192.HK), Tengsheng Bo medicine2137.HK), Conoco (2162.HK), Chaogju Eye Hospital2219.HK), Guichuangtong Bridge2190.HK), and Huang Medicine0013.HK), Keji Pharmaceutical2171.HK), Zhaoke Eye Hospital6622.HK), Nature Medicine (UPC.NASDAQ), SinoBiologics (6600.HK), Zhaoyan New Drugs6127.HK), Novo Nordisk Health (6606.HK), Tianyan PharmaceuticalADAG.NASDAQ), Beikang Medical (2170.HK), Jianbaimiao Miao Miao2161.HK), Minimally Invasive Cardiac Angioplasty (2160.HK), Ruili Medical Beauty2135.HK), Jiake Pharmaceutical1167.HK), and Bopan Medicine2142.HK), JD Health6618.HK), Deqi Medicine6996.HK), Rongchang Biology9995.HK), WuXi AppTec Genomics (2126.HK), Sinobiopharmaceuticals2096.HK), Yunding Xinyao1952.HK), Harvest Biotech6998.HK), ZaiDi Pharma9688.HK), Ocumivir1477.HK), Yongtai Biology6978.HK), Hype Pharma9989.HK), Kechuang Pharmaceutical9939.HK), Peijia Medical (9996.HK), Kangfang Biology9926.HK), Novo Nordisk China (9969.HK), Celestial BeingsIMAB.NASDAQ), Kanglonghua Cheng3759.HK), China Antibody3681.HK), Dongyao Pharmaceutical1875.HK), Yasheng Medicine6855.HK), Fosun Pharma (2696.HK), Hansoh Pharmaceutical3692.HK), Mabotech2181.HK), Founder Holdings1521.HK), Viva Biology1873.HK), CStone Pharmaceuticals2616.HK), Junshi Biology1877.HK), WuXi AppTec (2359.HK), Innovent Biologics1801.HK), Hualing Medicine2552.HK), BeiGene (6160.HK), Geely Pharma1672.HK), WuXi AppTec Biologics2269.HK), China Resources Medicine3320.HK), Yakult Scientific & Pharmaceutical2633.HK), and Huangzhong China MedicineHCM.NASDAQ), Biotechnology1548.HK),BBILife Sciences1035.HK) and so on. In terms of the number of submissions, Frost & Sullivan's healthcare team in Hong Kong is involved inIPO listingMaintain an absolute leading position,2018till2021Year after year, it has always ranked first in market share.
since2019year7Since the listing of the first batch of companies on the Sci-tech Innovation Board, Frost & Sullivan reports have also been widely referenced in the prospectuses of leading Sci-tech Innovation Board-listed companies, including: Yifang Biotechnology (688382.SH), Jicui YAKANG688046.SH), Haichuang Pharmaceutical688302.SH), Rongchang Biology688331.SH), Rendu Biology688193.SH), Capital Medical Group Holdings (688197.SH), and Yuanbiotechnology (688238.SH), YASIN Security688225.SH), Xidiwei688173.SH), MyWay Biotechnology688062.SH), Yarong Medicine688176.SH), BeiGene (688235.SH), Jiahemeikang688246.SH), DiZhe Medicine688192.SH), Novozymes688105.SH), Chengda Biology688739.SH), Gecko Micro688728.SH), Huaxi Biology688363.SH), Junshi Biology688180.SH), Zhejiang Genomics & Therapeutics Co., Ltd.688266.SH), Bai'ao Tai688177.SH), SinoCell (688520.SH) and others are considered one of the most powerful, professional, and influential industry research institutions in the sector. We aim to work with enterprises to understand industry trends, seize development opportunities, jointly promote innovation and upgrading of China's big health industry, and build a healthy future.
Recommended Reading
01.Frost & Sullivan assisted Zhiyun Health in successfully going public in Hong Kong(9955.HK)
03.Frost & Sullivan helpsPrenetics Global LimitedSuccessfully listed in the US(NASDAQ: PRE)
04.Frost & Sullivan assisted YunKang Group in successfully going public in Hong Kong(2325.HK)
07.Frost & Sullivan helps Clear Medical successfully go public in Hong Kong(1406.HK)
13.Frost & Sullivan assisted Yingpeng Technology in successfully going public in Hong Kong(2251.HK)
16.Frost & Sullivan assisted Yuhuan in successfully listing on the Hong Kong Stock Exchange(2256.HK)
17.Frost & Sullivan assisted Kunbo Medical in successfully going public in Hong Kong(2216.HK)
18. Frost & Sullivan assisted Xianruida in successfully going public in Hong Kong(6669.HK)
19.Frost & Sullivan assisted Kangsheng Global in successfully going public in Hong Kong(9960.HK)
21.Frost & Sullivan assisted ConocoAL on its successful listing in Hong Kong(2162.HK)
24.Frost & Sullivan assisted Hualong Medicine in successfully going public in Hong Kong(0013.HK)
26.Frost & Sullivan assisted Zhaoke Ophthalmology in successfully going public in Hong Kong(6622.HK)
27.Frost & Sullivan assisted Nature Pharma in successfully going public in the US(UPC.NASDAQ)
32.Frost & Sullivan assisted Jianbimiao Miao in successfully going public in Hong Kong(2161.HK)
34.Frost & Sullivan assisted RuiLi Medical Beauty in successfully going public in Hong Kong(2135.HK)
39.Frost & Sullivan assisted Rongchang Biology in successfully going public in Hong Kong(9995.HK)
43.Frost & Sullivan assisted Jiahé Biotech in successfully going public in Hong Kong(6998.HK)
45.Frost & Sullivan assisted OcularVista in successfully going public in Hong Kong(1477.HK)
47.Frost & Sullivan assisted Hype Pharma in successfully going public in Hong Kong(9989.HK)
49.Frost & Sullivan assisted Peijia Medical in successfully going public in Hong Kong(9996.HK)
50.Frost & Sullivan assisted Kangfang Biotech in successfully going public in Hong Kong(9926.HK)
52.Frost & Sullivan assisted Tianjing Biology in successfully going public in the US(IMAB.NASDAQ)
57.Frost & Sullivan assisted Fuhong Hanlin in successfully going public in Hong Kong(2696.HK)
61.Frost & Sullivan assisted ViaBio in successfully going public in Hong Kong(1873.HK)
64.Frost & Sullivan assisted WuXi AppTec in successfully going public in Hong Kong(2359.HK)

