Frost & Sullivan, in collaboration with Jintiancheng, Haitong Securities and LeadLeo, jointly releasedWhite Paper on the Trend and Strategies of Chinese Innovative Drugs and Medical Devices Going Global in 2024
Frost & Sullivan' Frost & Sullivan May 6, 2024, 10:18 shanghai
Contact number|021-5407-5836
Contact Email|PR@frostchina.com

With the growing prosperity of the global biopharmaceutical market and rapid technological progress, Chinese medical products have emerged on the international stage with their high cost-effectiveness and unique technical advantages. Strong government support and globalization trends have created unprecedented favorable conditions for domestic medical products to go global. Among them, the export of drugsThe License-out model dominates, while the overseas expansion of medical devices relies more on registration or acquisition models.
In recent years, Chinese pharmaceutical companies have actively integrated into the global pharmaceutical industryThe BD transaction boom has not only driven the research and development process of innovative drugs but also accelerated the global sharing and maximization of technology value. In 2023, China's innovative drugs and medical devices have seen a high-profile moment of going global. Many local innovative pharmaceutical companies have licensed their products to international pharmaceutical giants at highly competitive prices, with both the volume and amount of License-out transactions reaching new highs; at the same time, several domestic drugs have successfully obtained marketing approvals from overseas regulatory bodies such as the FDA. The globalization of medical devices has also reached new milestones, marking an acceleration of the internationalization pace of China's medical device industry.

Recently, Frost & SullivanFrost & Sullivan, in collaboration with LeadLeo and the leading venture capital firms in Jintiancheng, Haitong Securities and Suzhou Industrial Park, jointly hosted the 'Ocean Plan' - an industry summit forum on opportunities and challenges for domestic innovative drugs and medical devices to go global. At the forum, Frost & Sullivan, LeadLeo, Jintiancheng and Haitong Securities jointly released the highly anticipated 'White Paper on the Trends and Strategies of Chinese Innovative Drugs and Medical Devices Going Global in 2024' (hereinafter referred to as the 'White Paper'), which conducted an in-depth discussion on topics such as BD transaction-related situations, strategies and experiences, as well as legal risks during the process of going global.

The release ceremony of the 'White Paper' was presided over by Chen Yihui from the Shanghai branch of Industrial Bank Co., Ltd. Yang Xiaocheng, Partner and Managing Director of Frost & Sullivan Greater China, Zhang Zihui, Co-Chief of the Healthcare Group at Haitong Securities, and Wu Xuri, Partner at Jintiancheng, served as the initiators for the release of the 'White Paper'. Zhang Zihui, Co-Chief of the Healthcare Group at Haitong Securities, made a brief introduction to the content of the white paper on site.

Scan the QR code to get the report now.
Main Contents of the White Paper
1
Global healthcare market potential
The global medical market shows tremendous potential, with strong demand in developed countries and rapid growth in emerging markets. Chinese medical enterprises are actively going global, seizing opportunities through different models and addressing challenges. Developed countries have high health expenditures, bringing market opportunities for Chinese biopharmaceutical companies; emerging markets have great growth potential, accelerating market access for Chinese enterprises. Going global can enhance the technical level and brand image of Chinese medical enterprises and help them cope with domestic competition. Against the backdrop of globalization, Chinese medical enterprises need to actively embrace international markets for broader development.
2
Current Situation and Outlook of BD Transactions
01 globalBD License Transaction Overview
Global MedicineBD licensing transactions remain stable, with License-out becoming the mainstream choice for pharmaceutical companies going global, aiding in the international development of the pharmaceutical industry. Despite facing the challenge of the 'patent cliff', multinational pharmaceutical companies are still actively seeking high-quality external projects to cope with future profit pressures. From 2019 to 2023, the number and total amount of BD transactions in the global biopharmaceutical field remained stable, with relatively small fluctuations in upfront payments. At the 42nd JPMorgan Chase Healthcare Summit, several mergers and acquisitions and BD transactions were concluded, mainly involving areas such as AI pharmaceuticals, RNAi, and ADCs, and most are in the early research stage. Leaders of major well-known pharmaceutical companies have varying transaction attitudes and directions due to different corporate conditions and market judgments. In the future, with market demand and technological progress, global pharmaceutical BD transactions are expected to continue growing.
02 chinaBD License Transaction Overview
In 2023, China's pharmaceutical BD licensing transactions were active, showing several significant trends. The number and amount of license-out transactions increased significantly, surpassing those of license-in for the first time, indicating that China is becoming a global exporter of biopharmaceutical innovation pipelines. Domestic transactions quickly warmed up after a setback but still lagged behind foreign transactions due to regional equity and the financial strength of buyers.
variousBD transactions have their own characteristics. License-in focuses on introducing advanced foreign technologies and mature products, while License-out prefers earlier-stage projects. Domestic transactions emphasize clinical certainty more. In terms of transaction methods, domestic-to-domestic transfers tend towards a risk-sharing cooperation model, reflecting a cautious attitude towards unknown clinical risks.
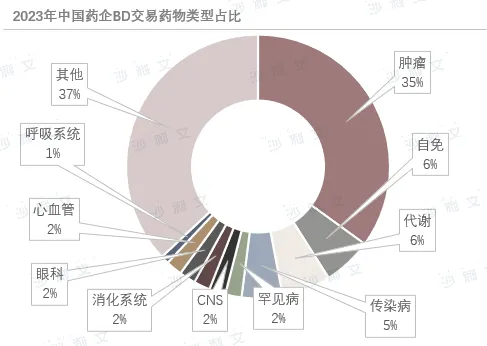
Source: Frost & Sullivan report
In niche segments,ADCs, macromolecular drugs, and small molecule drugs remain hotspots for transactions. At the same time, transactions in areas such as autoimmunity and infectious diseases are also increasing to meet diverse clinical needs. Among the top 10 BD transactions in 2023, license-outs dominated, with ADC transactions being particularly prominent, highlighting the innovative advantages of Chinese pharmaceutical companies in overseas transactions and the ADC field.
However, high-quality domestic buyers are still scarce, and transaction volumes are mainly driven byMNC dominance. In terms of projects, domestic sellers such as Mindray Biotech are favored by international MNCs, but the international commercialization capabilities of large domestic pharmaceutical companies still seem insufficient. This contradiction has led to a situation where although Chinese biotech product research has gone global, domestic buyers have limited influence in the international market.
In conclusion, Chinese medicineThe BD licensing transaction has shown a vigorous development trend, but it also faces some challenges. In the future, enhancing the international operation capabilities of domestic buyers and strengthening integration with the international market will be important development directions for China's pharmaceutical industry.
03 pastAnalysis of the Fulfillment Status of BD Transaction Milestones
Past pharmaceuticalsThe fulfillment of BD transaction milestones has been poor, with milestone payments worth $7.5 billion currently due but received by pharmaceutical companies in less than $1 billion. The high risk and investment required for new drug research and development, along with the uncertainty of clinical data, have led multinational companies (MNCs) to often choose to stop losses in a timely manner and terminate collaborations. Among the major transactions terminated over the years, many were related to poor product data, unclear commercial prospects, or adjustments in company strategic planning. This indicates that pharmaceutical BD transactions require careful risk assessment and flexible strategy adjustments to cope with market changes.
04 Domestic potentialBD trading target
Domestic pharmaceuticalsThe BD transaction field contains rich opportunities. New technologies, unmet consumer healthcare needs, and areas in urgent clinical drug demand will be key focuses for future BD transactions. In the field of central nervous system diseases, research and development of Alzheimer's disease and pain medications are particularly noteworthy, with the emergence of new drugs such as Lecanemab and VX-548 bringing new hope to the market. In the oncology field, competition for ADC and RDC drug research and development is fierce, but there are still projects with excellent medicinal properties worth looking forward to. In areas such as metabolic diseases like diabetes, obesity, and gout, there is an urgent demand for new drugs, especially for GLP-1/GIP dual-target drugs and gout treatment drugs. In addition, emerging fields such as small nucleic acid drugs and gene editing drugs are attracting more capital and attention from multinational corporations due to their unique advantages and potential. The development of these areas will provide a large number of opportunities for domestic pharmaceutical BD transactions, while also placing higher demands on R&D capabilities and market insight.
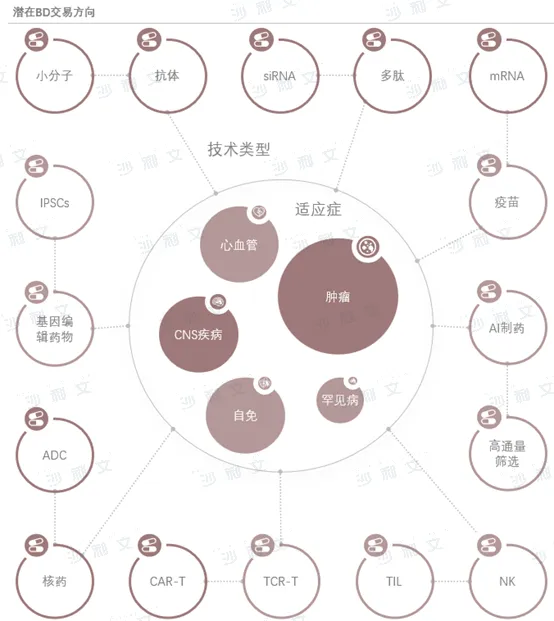
Source: Frost & Sullivan report
The White Paper provides global and ChinaAn overview of BD transactions is presented, which delves into the scale, structure, main participants, and transaction models. An in-depth analysis is conducted on the fulfillment of past BD transaction milestones, revealing potential domestic BD transaction targets including central nervous system diseases, tumors, metabolic diseases, and other areas. For more detailed content, please refer to the full report.
3
Research on the Overseas Market of Chinese Innovative Drugs
01 Analysis of Overseas Pharmaceutical Market Demand
The global pharmaceutical market size continues to grow, with an estimatedThe market will reach $2 trillion in 2027. The chemical drug market dominates, with a significant growth rate in the biopharmaceutical market, becoming an important growth point. Developed countries such as the United States and Europe lead the global pharmaceutical market, while emerging markets like China have great potential. The clinical pipeline is mainly composed of traditional drugs, but innovative therapies are constantly emerging, with oncology and immunology areas being global research and development hotspots. In the future, the pharmaceutical market will continue to grow, with more attention paid to innovative therapies and precision medicine.
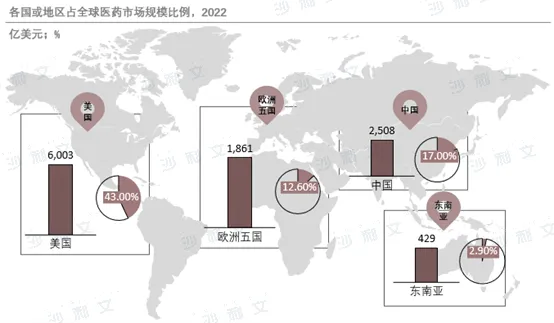
Source: Frost & Sullivan report
02 Analysis of the Current Situation of Chinese Pharmaceutical Companies Going Global
The overseas expansion of Chinese pharmaceutical companies has gradually expanded from bulk drugs to formulations, and then to innovative drugs. As the global focus on bulk drug production shifts to developing countries, China has risen as the world's largest bulk drug manufacturing base due to its cost advantages and R&D capabilities. In recent years, Chinese pharmaceutical companies have accelerated the internationalization process of innovative drugs through three models: leveraging other companies' overseas operations, joint ventures, and independent overseas development.
Although the 'borrowing a boat to go out to sea' model is flexible and requires low investment, enterprises may lose their voice in R&D and commercialization in overseas markets; the 'joint venture' model helps reduce policy barriers but cross-cultural communication is a major challenge; the 'self-sourcing' model allows for full control of the process but carries high risks and requires significant investment. With the enhancement of pharmaceutical companies' R&D innovation capabilities and intensified international competition, joint ventures and self-sourcing models are gradually gaining attention.
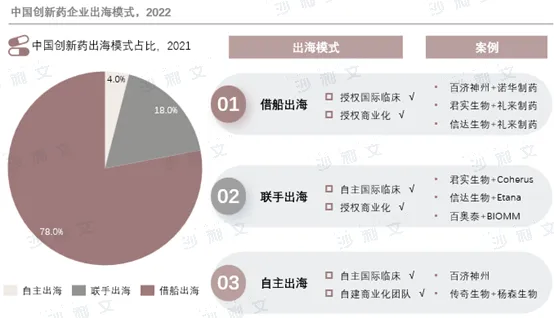
Source: Frost & Sullivan report
Although Chinese pharmaceutical companies mainly focus on the domestic market, they have insufficient overseas expansion. Structurally, biotech companies that emphasize overseas clinical trials and product commercialization have a higher proportion of overseas revenue. Despite China's significant progress in the field of innovative drugs, its proportion of overseas revenue is still lower than that of international peers. Leading biotech companies such as BeiGene achieve a high proportion of overseas revenue through licensing transactions or drug sales.
Overseas clinical trials are mainly concentrated in the United States, with multi-center trials gradually attracting attention. The efficiency of drug registration and cost minimization drive clinical trials in countries such as Australia. In terms of overseas product pipeline layout, leading companies such as BeiGene and Hengrui Medicine have rich overseas pipelines and are expected to become the focus of future commercialization.
From 2016 to 2022, the number of License-outs by Chinese pharmaceutical companies increased significantly, mainly focusing on anti-tumor and anti-infection areas, with antibody drugs being the main focus. The transaction amount exceeded $40 billion, and there was active trading in the fields of bispecific antibodies (bAb) and antibody-drug conjugates (ADCs), indicating an acceleration in the internationalization of Chinese innovative pharmaceutical companies. However, target selection is still concentrated in hot areas, leading to fierce competition on the internationalization track.
Looking ahead, Chinese pharmaceutical companies need to deepen international cooperation, expand overseas markets, and at the same time strengthen innovation and research and development to enhance their core competitiveness, so as to achieve broader international development. This is a challenging path, but it is also a crucial step for China's pharmaceutical industry to achieve global layout and improve international competitiveness.
The White Paper sorts out the current situation of Chinese pharmaceutical companies going global, including their overseas journey, models, market focus, overseas clinical trials and product pipeline layout, as well asFor more details on license-out transactions, please refer to the full report.
03 Opportunities and Challenges for Chinese Pharmaceutical Companies Going Global
Chinese pharmaceutical companies have made remarkable progress in the field of innovative drugs, but compared with developed countries, there is still a significant gap in drug prices, providing a huge market space for Chinese pharmaceutical companies to go global. With the continuous improvement of innovation levels, Chinese pharmaceutical companies areModels such as License-out have gone global and gradually gained recognition in international markets. However, the pressure of domestic medical insurance negotiations for price cuts and the difficulties in increasing sales of innovative drugs have left pharmaceutical companies facing a dilemma of declining capital market valuations. Against this backdrop, going global has become a key strategy for Chinese pharmaceutical companies to digest supply and earn market returns.
The international integration of the regulatory system and the improvement of approval processes have provided strong support for Chinese pharmaceutical companies going global. At the same time,The enhancement of CXO enterprises' strength and the accumulation of international experience have further promoted the process of new drug research, development, and commercialization by Chinese pharmaceutical companies going global. However, in the process of going global, Chinese pharmaceutical companies also face some challenges, such as relying on local clinical data for research and development thinking, and adapting to market demands in different countries.
To avoid obstacles in going global, Chinese pharmaceutical companies should transform their R&D mindset, ensure detailed experimental data, and fully consider the prevalence of diseases in overseas markets. At the same time, they should explore niche indications and the demand for drugs for small-batch cancer patients, enhancing their overseas competitiveness through differentiated drug advantages. Looking ahead, with the continuous improvement of innovation capabilities and the accumulation of overseas experience, Chinese pharmaceutical companies are expected to play an increasingly important role in the global pharmaceutical industry.
The 'White Paper' sorts out the opportunities and challenges for Chinese pharmaceutical companies going global, pointing out that while they face opportunities such as large market space and improved innovation levels, they also face challenges like price pressure and a shift in R&D mindset. For more detailed content, please refer to the full report.
4
Research on the Overseas Market of Innovative Medical Devices in China
01 Analysis of Overseas Medical Device Market Demand
The global medical device market is growing steadily, led mainly by the US and Europe, but the markets in Asia-Pacific and developing countries hold great potential. The scale of China's medical device market has expanded rapidly, and going global has become a key strategy for enhancing competitiveness. In the face of high-quality requirements from overseas markets, Chinese enterprises need to continuously improve their technical level. At the same time, the industrial advantages of countries such as the UK, France, and Germany complement those of China, and cooperation opportunities are also emerging in markets in Latin America and Southeast Asia. With the help ofCooperation frameworks such as the Belt and Road Initiative and RCEP enable Chinese enterprises to further expand overseas markets and achieve international development.
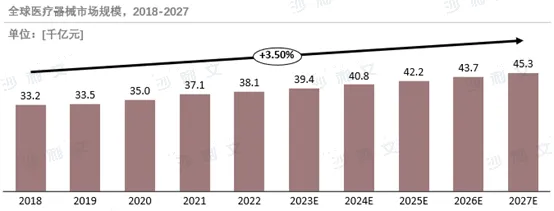
Source: Frost & Sullivan report
02 Analysis of the Current Situation of Chinese Medical Device Enterprises Going Global
China's medical device exports have gone through more than three decades, starting from low-value consumables and gradually expanding to high-end technologies and products, with continuous enhancement of international competitiveness. In recent years, although export amounts have declined due to the impact of the pandemic, the stable supply capacity of enterprises has won global recognition, and the export scale still exceeded expectations.$20 billion. The number of overseas registrations for Chinese medical devices continues to grow, but they face challenges in registration regulations. The export market is mainly Europe and America, with an increasingly rich variety of products. Chinese medical device companies have seen significant growth in their overseas business, but compared to international peers, they still have a gap in the proportion of international revenue. There is considerable room for improvement in the future, requiring continued strengthening of R&D capabilities, enhancement of product quality and service levels, and further expansion of overseas markets.
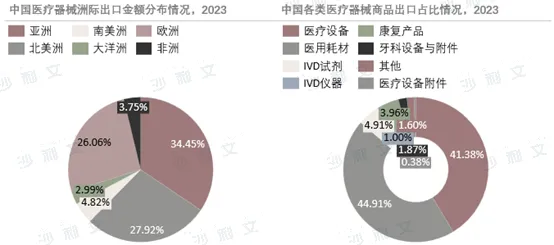
Source: Frost & Sullivan report
03 Analysis of Sub-sectors for Chinese Medical Devices Going Global
The overseas markets for medical devices in China are characterized by different segments. Low-value consumables, as the core of exports, have expanded overseas markets with their cost advantage and supply capabilities, especially during the pandemic, which is expected to further increase their global market share. The total export value of in vitro diagnostic products has been growing year by year, and domestic manufacturers have enhanced their competitiveness, demonstrating a diversified overseas strategy. High-value consumables face challenges in product research and development and sales models when going global, but their clinical and technical advantages have become the core competitiveness, with huge potential in overseas markets. In the field of medical equipment, although the overseas market for high-end equipment such as imaging and surgical robots is still limited, some products have reached high-end levels and occupied market shares. As domestic enterprises gradually replace imported shares, tapping into overseas market potential has become an inevitable choice.
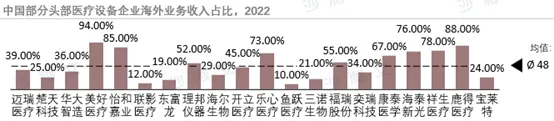
Source: Frost & Sullivan report
04 Opportunities and Challenges for Chinese Medical Device Enterprises Going Global
The overseas medical device market has high pricing, providing opportunities for Chinese enterprises to go global. However, domestic volume-based procurement policies have led to significant price drops in medical devices, squeezing profit margins for enterprises. Therefore, Chinese medical device companies are seeking overseas markets to find new growth points. However, there are significant differences in the regulatory requirements for medical device registration across countries, especially in the US and Europe, which require high-standard review and certification. At the same time, international market competition is fierce, and Chinese companies need to strengthen their brand influence and market competitiveness. In recent years, the innovation momentum in China's medical device industry has been strong, with many products possessing global competitiveness, providing new opportunities for overseas business. However, choosing an overseas sales model is also a major challenge, requiring a balance of the advantages and disadvantages of distribution models and direct sales models, and formulating market expansion strategies suitable for their own development. Facing challenges and opportunities, Chinese medical device companies need to continuously enhance their technical strength, adapt to different market demands, and achieve international development.
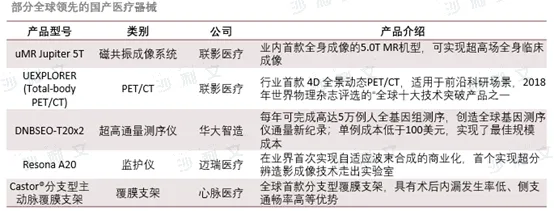
Source: Frost & Sullivan report
The White Paper analyzes the overseas markets for innovative medical devices in China, covering overseas demand, current situation of enterprises going global, analysis of niche markets (listing four specific niche markets), as well as the opportunities and challenges faced. For more detailed content, please refer to the full report.
5
Overview of Laws and Regulations Governing Overseas Transactions of Drugs and Medical Devices
01 Overview of Laws and Regulations Governing Overseas Transactions of Pharmaceuticals and Medical Devices
The transaction of pharmaceutical and medical device technology licenses, especially cross-border transactions, involves a complex legal and regulatory system. Both parties involved in the transaction need to comply with basic laws such as the Civil Code and Patent Law, while also adhering to the professional regulatory regulations of the pharmaceutical and medical device industry to ensure compliance throughout the entire lifecycle. There are also strict regulations regarding taxation, technology import and export, and cross-border data transmission. In addition, the laws of the other country and international treaties must be considered. To ensure the smooth progress of the transaction, both parties should fully understand and comply with relevant laws and regulations to avoid legal risks.
02 Main legal issues in pharmaceutical and medical device overseas transactions
The overseas transaction of pharmaceuticals and medical devices involves multiple key legal issues. The transaction contract needs to specify licensing terms, fee allocation, milestone payments, and sales distribution mechanisms. The licensor should ensure they own complete intellectual property rights and avoid infringement. The contract should define the difference between intellectual property transfer and licensing, protect intellectual property rights, clarify jurisdiction and choice of law. At the same time, it is necessary to consider the impact of third-party rights, competition law, and anti-monopoly law. The contract should include a breach of contract and dispute resolution mechanism to protect the interests of both parties. The introduction of a joint steering committee can coordinate cooperation details and deal with emergencies. In terms of anti-corruption compliance, all parties need to comply with domestic and international anti-bribery regulations and may agree in the transaction documents to follow the licensor's management system. At the same time, attention should be paid to cross-border transmission of sensitive personal information and human genetic resource information, comply with national regulatory systems, and fulfill approval or filing procedures. In addition, attention also needs to be paid to pharmaceuticals and medical devices.Transaction details such as MAH clauses, production and supply, commercial arrangements, as well as core legal issues like technology import and export controls and tax supervision.
03 Key Points of Intellectual Property Concerns in Overseas Transactions of Pharmaceuticals and Medical Devices
In the overseas transaction of pharmaceuticals and medical devices, intellectual property rights are a core concern. Firstly, it is necessary to clarify the ownership of intellectual property rights to avoid subsequent disputes; secondly, define licensing rights and ensure the rights and interests of both parties; at the same time, pay attention to the layout and compliance issues of intellectual property rights, conduct patent freedom-to-operate analysis, and reduce the risk of infringement. In addition, tax issues related to intellectual property rights also need to be considered to ensure tax compliance. Finally, establish a dispute resolution mechanism for intellectual property rights to provide legal protection for transactions. Considering these key points comprehensively helps to reduce the risks of intellectual property rights in overseas transactions of pharmaceuticals and medical devices.
The 'White Paper' sorts out the legal and regulatory system involved in pharmaceutical and medical device overseas transactions, key legal issues, and points of attention regarding intellectual property rights. It discusses the basic laws and industry regulatory regulations that need to be complied with, and pays attention to core issues such as contract terms, intellectual property ownership, and licensing rights. For more detailed content, please refer to the full report.
Highlights of the Conference
The first half of this forum was presented byYu Zhengchun, Senior Partner at JintianchengThe lawyer served as the moderator.

Guiding Unit of This Event,He Jia, Deputy Director of the Suzhou Industrial Park Enterprise Development Service CenterA speech was delivered, with opening remarks made by Gao Gehui, a senior partner at Jintiancheng and a lawyer at the firm.


Shen Sheng, Director of the Healthcare Group at Haitong SecuritiesPublished a paper titled 'Global and Domestic BiomedicineThe keynote speech of the 'BD Transaction Tracking Report' introduced the global and domestic BD licensing transaction overview, analyzed past BD transaction milestone achievements, potential domestic BD transaction targets, and the development trends of BD transactions for medical device companies.

Huang Haoyu, former executive of Roche and Takeda Pharmaceutical, former general manager of XinnorinCentered onThe speaker shared on the theme of 'MNC M&A Trends and Transaction Practices'. First, they reviewed typical domestic biotech mergers and acquisitions transactions over the past year, analyzed the trend of overseas MNC mergers and acquisitions of domestic biotech companies, and from the perspective of the acquired party, discussed key issues in the transaction practices of domestic biotech company mergers and acquisitions.

Hangzhou HEGYA Biopharmaceutical Co., Ltd.CEO GuantaoWithA speech titled "BD Transaction Negotiation Strategies and Experience Sharing" was delivered, which provided an in-depth interpretation from aspects such as the value assessment logic of innovative drugs, new drug research and development based on a value system, BD transaction negotiation strategies, and skills.

The roundtable session followed,Hu Xuan, Director of the Healthcare Group at Haitong SecuritiesThe panelists participating in the roundtable discussion includeSenior Advisor to Strategic Development, Senior Advisor to Warburg Pincus Investment in the US, Former Vice President of Eli Lilly China,Chen Hongbo, Vice President of GSK Global,Gu Leming, former executive at Johnson & Johnson and Philips China, current partner at Yuansheng Venture Capital,Dr. Liu Qian, Shanghai Wish Biotechnology Co., Ltd.,Dr. Wang Yiyi, Co-founder of Wuxi Zhikang Hongyi Biotechnology Co., Ltd.,Jiangsu Famaisheng Medical Technology Co., Ltd.VP Chen Qixun,Zhang Wei, Director and General Manager of Dite Medical Technology (Suzhou) Co., Ltd.,Xu Yuxin, Investment Director of SDIC China Merchants Investment Management Co., LtdThe guests engaged in an in-depth exchange on topics such as the considerations for pharmaceutical and medical device companies' outbound strategies, discussions on regional advantages, and suggestions to avoid pitfalls.

After the tea break, the forum entered the second half of its agenda, which was presented byJintiancheng Partner Wu XuriThe lawyer served as the moderator.

Huang Jingjing, Senior Partner at JintianchengFirst, bring inThe sharing of 'Compliance Issues in MNC Mergers and Acquisitions of Domestic Biotech Enterprises' lists cases and key points of MNC mergers and acquisitions of domestic Biotech enterprises, and provides compliance suggestions such as emphasizing daily compliance, timely communication, and teamwork.

Subsequently,Ding Yijü, Head of M&A Department at Haitong Bank Macau BranchJiao delivered a speech on the topic of 'Overseas M&A Transaction Process and Risk Control', explaining the cross-border M&A process. Combining practical transaction cases, he focused on analyzing project development, due diligence, agreement negotiation and main transaction documents, external regulatory approvals, Price adjustment mechanism.

The second roundtable session begins.Lu Xuezhong, Senior Partner at JintianchengThe lawyer chaired the discussion of this group, and the roundtable was invited toWang Zhe, Director of the Healthcare Group at Haitong Securities,Liu Tingting, a senior lawyer at Jintiancheng,Liu Donghai, Director of PWC's Corporate Financing and M&A Services,Yaotang (Shanghai) Biotechnology Co., Ltd.Director BD and PR, Lin Jian,Chen Haosi, Assistant General Manager of Xiyuan Anjian Medicine (Beijing) Co., Ltd.andDr. Cheng Ying, Director and Business Development Director of Shanghai Kezhou Pharmaceutical R&D Co., Ltd.The guests thenThe focus on BD transaction negotiation and transaction clause design, legal and other related risk prevention are comprehensively discussed.

Finally, byZhou Han, Director and Senior Partner of the Management Committee of Jintiancheng Suzhou BranchThe lawyer delivered a closing speech. The event has come to a successful conclusion.

Recommended Reading

↓↓Long-press to scan the QR code below to obtain ↓↓

Frost & Sullivan
Global growth consulting firm, Frost & SullivanFrost & Sullivan, abbreviated as 'Frost & Sullivan', integrates 64 years of global consulting experience. Over the past 27 years, it has dedicated itself to serving the booming Chinese market with a global perspective, helping clients accelerate their business growth and achieve leading benchmarks in industry growth, innovation, and technology.
Contact number:021-5407-5836
Contact email:PR@frostchina.com
Copyright Notice
Thank you for your attention and support to Frost & Sullivan. If you wish to reprint articles from our WeChat official account, please:
·
Leave a message in the original text, indicating the name, introduction, and WeChat official account of the reprinting institution.ID;
·
·
Fill in the author on the editing page"Frost & Sullivan";
·
·
All reposts must not alter, misinterpret, interpret, or adapt the original text in any way. Frost & Sullivan reserves the copyright and the right to interpret all original articles. In case of violation, Frost & Sullivan reserves the right to pursue corresponding legal responsibilities according to the law.
·
About Frost & Sullivan
Global growth consulting firm, Frost & SullivanFrost & Sullivan, abbreviated as 'Frost & Sullivan', integrates 63 years of global consulting experience. Over the past 26 years, it has dedicated itself to serving the booming Chinese market with a global perspective. It has helped over 10,000 clients accelerate their business growth, assist them in achieving growth, technological innovation, and leading benchmarks within the industry, and realize capital operation goals such as financing and listing.
Frost & Sullivan has deep roots in the global capital markets and corporate consulting services, and its innovative propositions“Total Investment Management (TIM)” provides enterprises with comprehensive investment and financing as well as various other professional consulting services, including investment and financing CDD, valuation services, technology advisors, financial advisors, ESG, fundraising and investment feasibility studies, bond issuance industry advisors, industry advisors, assessment services, award services, industry white papers, strategic and management consulting, planning consulting, technical insights, etc.
Frost & Sullivan's investment and financing business in Greater China has achieved full industry coverage of the Chinese national economy, including a high level of attention to all economic hotspots such as the new economy and new infrastructure, covering digital infrastructure, consumer electronics, dual-carbon new energy, healthcare and life sciences, catering and new retail, semiconductors and integrated circuits, smart homes, automotive and mobility, health care services, food and beverages, information and communication technology, fintech, real estate and property, mining and smelting, beauty and fashion, big data and artificial intelligence, logistics and supply chain, construction technology and decoration, special new materials, culture and entertainment, enterprise-level services, cross-border e-commerce trade, infrastructure construction, environmental protection and energy-saving technology, education and training, etc.
Since the Frost & Sullivan team began providing investment and financing advisory services to corporate leaders and their management teams, it has helped nearly a thousand companies successfully list in Hong Kong and overseas, making it a leading enterprise in the field of domestic investment and financing strategy consulting. In recent years,For over 10 years, Frost & Sullivan has ranked first in market share among professional industry advisors for Chinese companies listing in Hong Kong and overseas; and in recent years, Frost & Sullivan reports have been widely cited in the prospectuses of leading A-share and Sci-Tech Innovation Board listed companies, primary and secondary market research reports, and other capital market disclosure documents.
For over 63 years, Frost & Sullivan has helped a large number of clients (including Fortune Global 1000 companies, top domestic and foreign financial institutions, and various other leading enterprises) achieve strategic goals by completing work including but not limited to due diligence, valuation analysis, and third-party evaluations through its nearly 50 offices around the world, utilizing a powerful database and expert pool, and applying rich professional knowledge and consulting tools; it has established a market position confirmation system and innovatively proposed the “FSBVFrost & Sullivan Brand Value Model,” providing market position confirmation and brand valuation services to over 500 enterprises, continuously helping many Chinese brands towards their growth future.


