Frost & Sullivan
Guangdong Dongyang Pharmaceutical Co., Ltd. (Stock Code: 6887.HK) successfully went public in Hong Kong on August 7, 2025. The company is engaged in the research, development, production, and commercialization of drugs, focusing on innovative medicines, as well as modified new drugs, generic drugs, and biosimilars. Currently, the company has established an industry-leading R&D platform, production facilities that meet international standards, and a global sales network. It strategically focuses on therapeutic areas such as infections, chronic diseases, and tumors, addressing major unmet medical needs. Frost & Sullivan (hereinafter referred to as 'Frost & Sullivan') provides exclusive industry advisory services for the listing of Guangdong Dongyang Pharmaceutical Co., Ltd., and we warmly congratulate them on their successful listing.
Guangdong Dongguangyang Pharmaceutical Co., Ltd. (hereinafter referred to as 'Dongguangyang Pharmaceutical')YuOn August 7, 2025, it was listed on the main board of the Hong Kong Stock Exchange under the ticker 'Dongguangyang Pharmaceutical' (6887.HK), becoming the first innovative case of 'H-share absorption merger privatization + introduction listing' on the Hong Kong Stock Exchange.
During the process of listing in Hong Kong this time, Frost & Sullivan mainly undertook the following tasks: helping the issuer accurately and objectively understand its positioning in the target market, using objective market data to discover, support and highlight the issuer's competitive advantages, assisting the issuer, investment banks and other intermediaries in completing the writing of relevant parts of the prospectus (such as overview, competitive advantages and strategy, industry overview, business and other important chapters), helping the issuer complete communication with the Hong Kong Stock Exchange and investors, assisting investors in quickly understanding the market ecosystem and competitive landscape, and providing feedback on various industry-related issues from the Hong Kong Stock Exchange to the issuer.
Frost & Sullivan has always been a leader in helping companies go public in Hong Kong. According to LiveReport's big data (statistical data as of June 30, 2025), from January to June 2025, as well as during the past 12 and 36 months, Frost & Sullivan provided listing industry advisory services for 29 (market share 71%), 60 (market share 67%), and 164 (market share 69%) Hong Kong stock IPOs, ranking first in terms of number. It has a wealth of industry experience and communication experience with regulatory authorities, exchanges, investment and financing institutions, and various related institutions.
PART/1
Investment Highlights
-
Dongguang Medicine has outstanding independent innovation capabilities. As of June 30, 2024, the company has been approved for a total of 147 drugs globally (including China, the United States, and Europe), with more than 100 ongoing projects, including 45 Class I innovative drugs. Among them, 3 are under review by the National Medical Products Administration, and 10 innovative drugs in development are in Phase II/III clinical trials. The company is committed to applying AI technology to all stages of drug research and development. Its AIDD laboratory has submitted the first candidate drug HEC169584 for the treatment of NASH, fully demonstrating its comprehensive strength in cutting-edge technologies and new drug research and development.
-
The company also holds a leading position in the production sector, with two major production bases located in Dongguan Songshan Lake and Hubei Yidu. The total area exceeds 1,300 acres and it has international first-class GMP certification. The Songshan Lake base has passed GMP certifications from China, the United States, and Europe, and has the capacity to produce 1.8 billion tablets/year of chemical drugs; the Yidu base is the world's largest oseltamivir production base, with its annual insulin production capacity expanded to 100 million vials in 2024. At the same time, the company is building a large-scale biopharmaceutical production base that is expected to be put into use in 2025, providing solid support for the commercialization of new drugs in the future.
-
In terms of sales channels, Dongguangyang Pharmaceutical has established a wide and deep national sales network, covering 32 provinces and cities across China, approximately 2,500 tertiary hospitals, 9,500 secondary hospitals, and 75,000 primary healthcare institutions. Overseas, the company has expanded to 8 countries and regions including the United States, Germany, and the United Kingdom, and has signed exclusive licensing agreements with international partners for multiple products, demonstrating its competitiveness in the global market. Kewei®Oseltamivir phosphate, as a core product of the company, accounted for 64.8% of the Chinese oseltamivir market and 50.5% of the overall anti-influenza drug market in 2023, validating its outstanding market execution and product recognition;
-
In recent years, Dongguangyang Pharmaceutical has ranked among the top in several authoritative pharmaceutical industry lists for many consecutive years. It has been included in the TOP 20 of the 'China's Comprehensive Strength Ranking of Pharmaceutical R&D' and is part of the first tier of the 'Top 100 Innovative Pharmaceutical Enterprises'. The company's industry influence continues to grow. In addition, the company has been active in medical insurance access. As of June 30, 2024, one of its innovative drugs has been included in the national medical insurance drug list.
PART/2
Overview of the Chinese Influenza Drug Market
Influenza, commonly known as 'flu', is an infectious disease with a higher incidence rate among children under 5 years old and the elderly. There are four types of influenza viruses, among which human influenza A and B viruses cause seasonal epidemics almost every winter. With the improvement of public health awareness and the country's emphasis on the prevention and control of infectious diseases, the demand for anti-influenza drugs has maintained a steady growth. Drugs represented by oseltamivir on the market have become first-line treatment medications and their clinical accessibility has been enhanced through the wide coverage of the national medical insurance catalog.
PART/3
Market Scale and Forecast of Anti-Influenza Drugs in China
The scale of the Chinese influenza drug market increased from RMB 32 billion in 2018 to RMB 44 billion in 2022, with a compound annual growth rate of 8.4%. Although the overall trend is upward, due to the impact of COVID-19 in 2020 and 2021, the number of influenza cases decreased, leading to a decline in market size. However, with the end of the COVID-19 pandemic, the number of cases of other respiratory infectious diseases increased significantly. In 2023, the impact of the influenza epidemic in China was broader and lasted longer than before the COVID-19 pandemic, and the market size increased significantly to RMB 110 billion in 2023. Unlike 2023, there were no severe influenza outbreaks in 2024, resulting in reduced demand for influenza drugs. Therefore, the market forecast for influenza drugs in China in 2024 is expected to decrease to RMB 8 billion. It is expected that the market size will recover by 2026 and reach RMB 110 billion, and further increase to RMB 116 billion by 2030.
China Influenza Antiviral Drugs Market, 2018 - 2030E
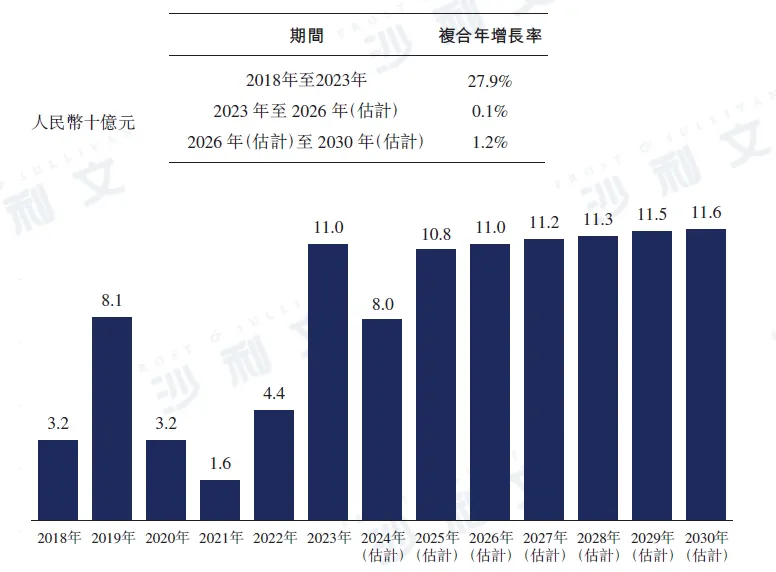
Note: Does not include traditional Chinese medicine
Data source: Analysis by Frost & Sullivan
PART/4
Market Scale and Forecast of Anti-Influenza Drugs in China
The scale of the Chinese influenza antiviral drug market was RMB 11 billion in 2023, among which the sales of oseltamivir phosphate reached RMB 8.6 billion in 2023, accounting for 78.0% of the total market share that year. In the same year, the sales of parainfluenza virus vaccine (PRADAX) were RMB 9.437 billion, accounting for 8.6% of the total market share. In 2023, Dongyang Pharmaceutical's Kexing®The sales volume of (Oseltamivir Phosphate) reached approximately RMB 5.5 billion, accounting for 50.5% of the Chinese influenza antiviral drug market and 64.8% of the entire Chinese oseltamivir phosphate market.
2023 China Influenza Antiviral Drug Market
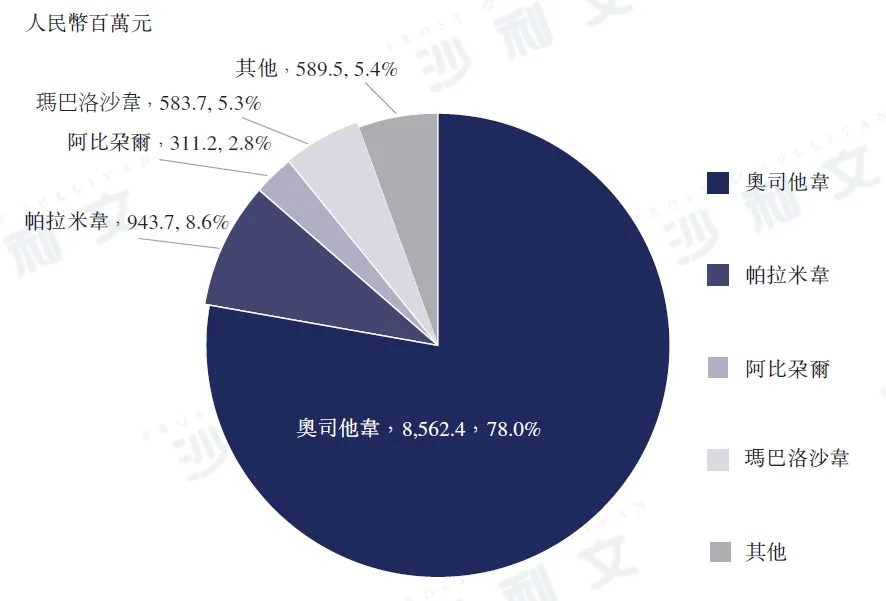
Data source: Analysis by Frost & Sullivan
2023 China Oseltamivir Phosphate Drug Market
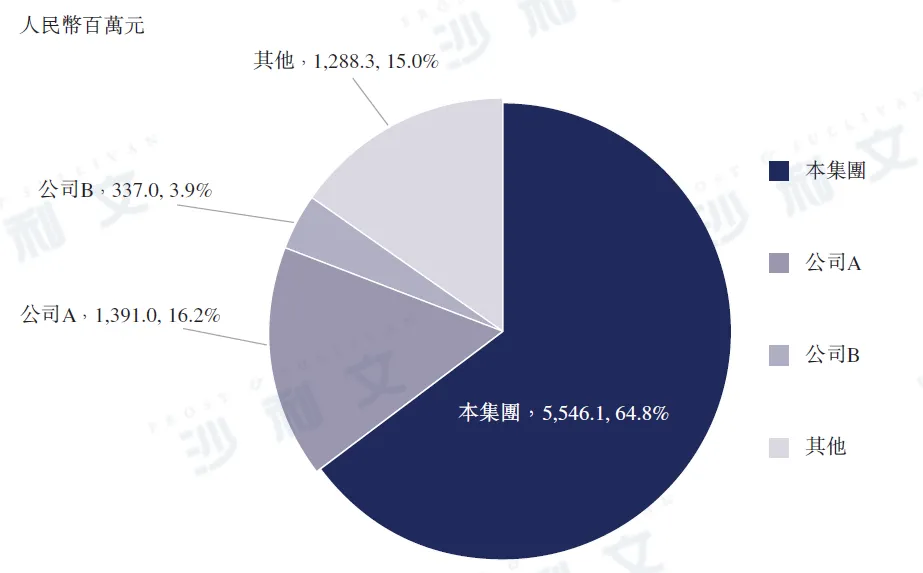
Note:
Company A is a listed multinational healthcare holding company headquartered in Switzerland, focusing on the development of drugs and diagnostics.
2. Company B is an A-share listed company headquartered in Guangzhou, focusing on the research, development, production, and sales of pharmaceutical products.
Data source: Analysis by Frost & Sullivan
PART/5
Overview of the Chinese Hepatitis Drug Market
Hepatitis B is an infectious disease characterized by liver inflammation caused by the hepatitis B virus (HBV). Its clinical symptoms include loss of appetite, liver pain, and physical weakness. HBV infection can be acute or chronic. After infection with HBV, it may develop into chronic hepatitis, which can progress to cirrhosis, liver failure, and hepatocellular carcinoma. The symptoms of chronic hepatitis C can also be difficult to identify in the early stages, with fatigue being the most common symptom, which can last for 20 to 25 years. Jaundice (yellowing of the skin or eyes) may indicate severe disease progression. Some patients with cirrhosis may also clinically develop hepatocellular carcinoma.
PART/6
The market scale and forecast of hepatitis drugs in China
In 2023, the market size of anti-HBV drugs in China reached RMB 9.9 billion. It is expected that the market size of anti-HBV drugs in China will continue to grow in the future, reaching RMB 16.7 billion by 2026 and further increasing to RMB 434 billion by 2030. The compound annual growth rate from 2023 to 2026 is expected to be 19.1%, while the compound annual growth rate from 2026 to 2030 is expected to reach 27.0%.
China's anti-HBV drug market, 2018 - 2030E
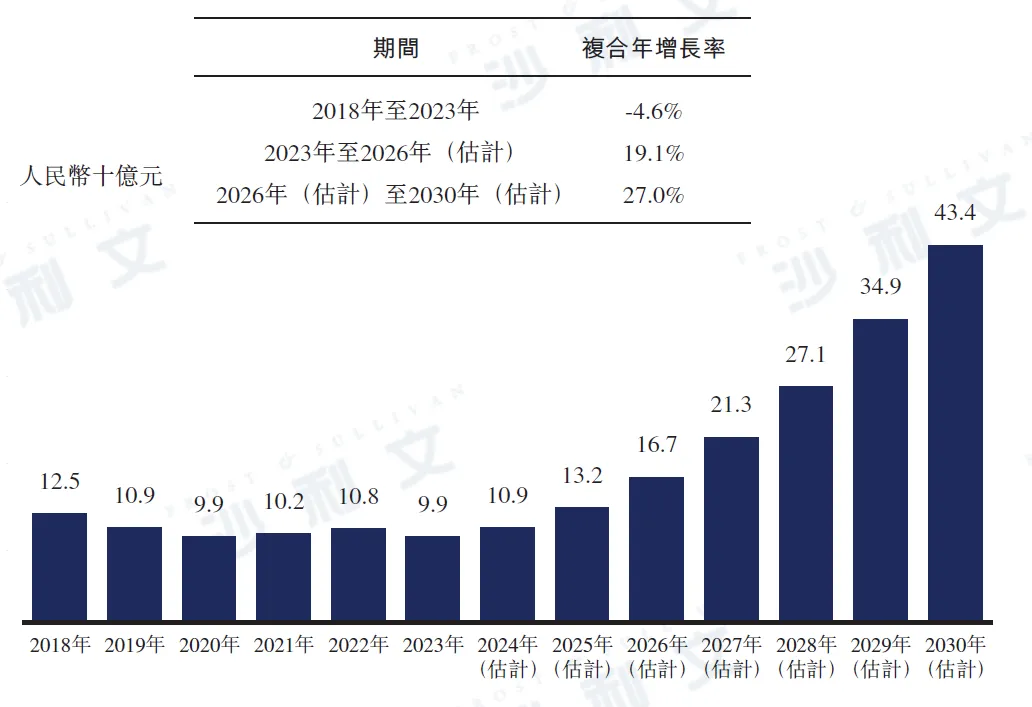
Data source: Analysis by Frost & Sullivan
In 2023, the market scale of anti-HCV drugs in China reached 3.7 billion yuan. Due to (i) DAA being included in the national medical insurance drug list in 2020 and the increasing number of DAA drugs approved in China, its penetration rate has been continuously rising; (ii) The National Health Commission of China and eight other government departments implemented the 'National Public Health Hazard Elimination Action Plan for Hepatitis C (2021-2030)' in 2021 (which includes an overall goal and 15 specific goals covering health education, comprehensive prevention interventions, testing, and treatment); and (iii) increased production capacity, the market for anti-HCV drugs in China will continue to grow. It is expected that the market scale of anti-HCV drugs in China will reach 4.5 billion yuan by 2026, further reaching 4.9 billion yuan by 2030. The compound annual growth rates from 2023 to 2026 and from 2026 to 2030 are expected to be 7.0% and 2.1%, respectively.
China Anti-HCV Drug Market, 2018 - 2030E
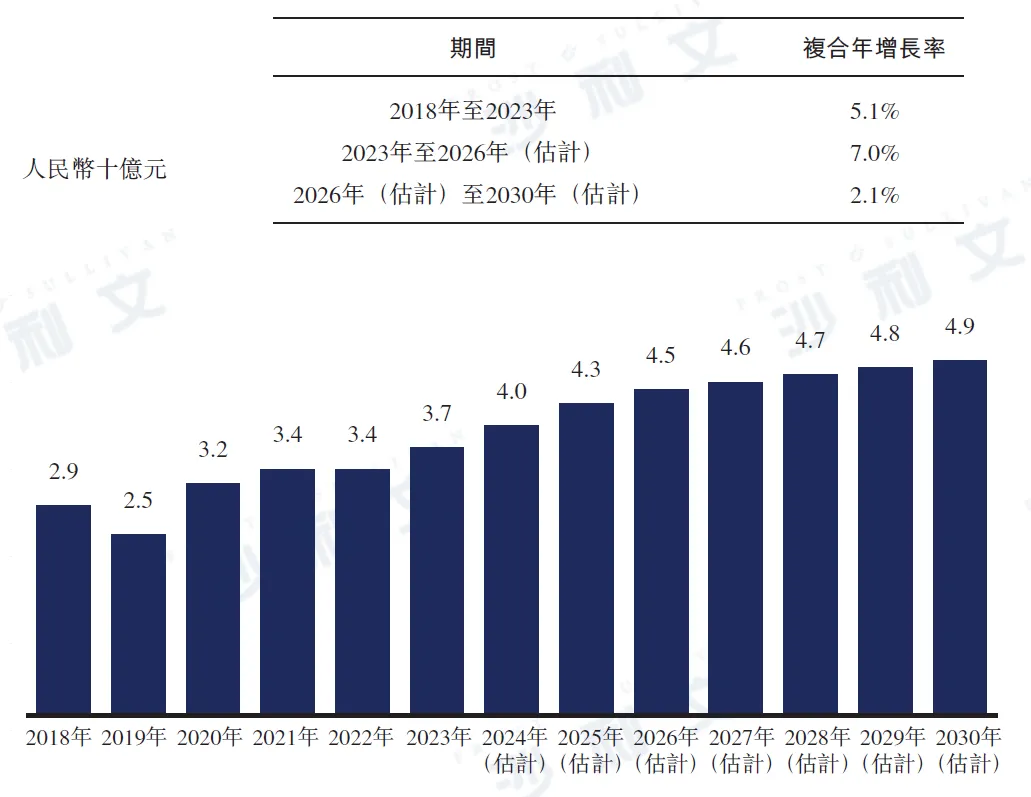
Data source: Analysis by Frost & Sullivan
PART/7
Overview of the Chinese Metabolic Disease Drug Market
The World Health Organization defines chronic diseases as persistent, usually slow-progressing, and non-infectious illnesses. Chronic diseases require long-term treatment, care, and special rehabilitation. They are characterized by a long duration of illness. With the improvement of living standards, chronic diseases have become the leading cause of death globally. Chronic diseases generally include metabolic diseases such as diabetes, chronic respiratory system diseases such as chronic obstructive pulmonary disease (COPD), and neuropsychiatric disorders. Problems caused by impaired metabolism can lead to metabolic diseases, affecting various tissues and organs throughout the body, as well as intelligence, growth and development, and mental state. The main risk factors include poor lifestyle habits, age, obesity, environmental pollutants, abnormal blood sugar levels, blood pressure, blood lipids, uric acid, etc. Patients may suffer from multiple complications or comorbidities, such as obesity, non-alcoholic fatty liver disease, hypertension, and abnormal blood lipids.
PART/8
Market Size and Forecast of Metabolic Disease Drugs in China
In 2023, the market size of metabolic disease drugs in China reached 1074 billion yuan, with a compound annual growth rate of 5.7% from 2018 to 2023. It is estimated that this market size will reach 1511 billion yuan in 2026, 2195 billion yuan in 2030, with a compound annual growth rate of 12.1% from 2023 to 2026, and 9.8% from 2026 to 2030.
China Metabolic Disease Drugs Market, 2018 - 2030E
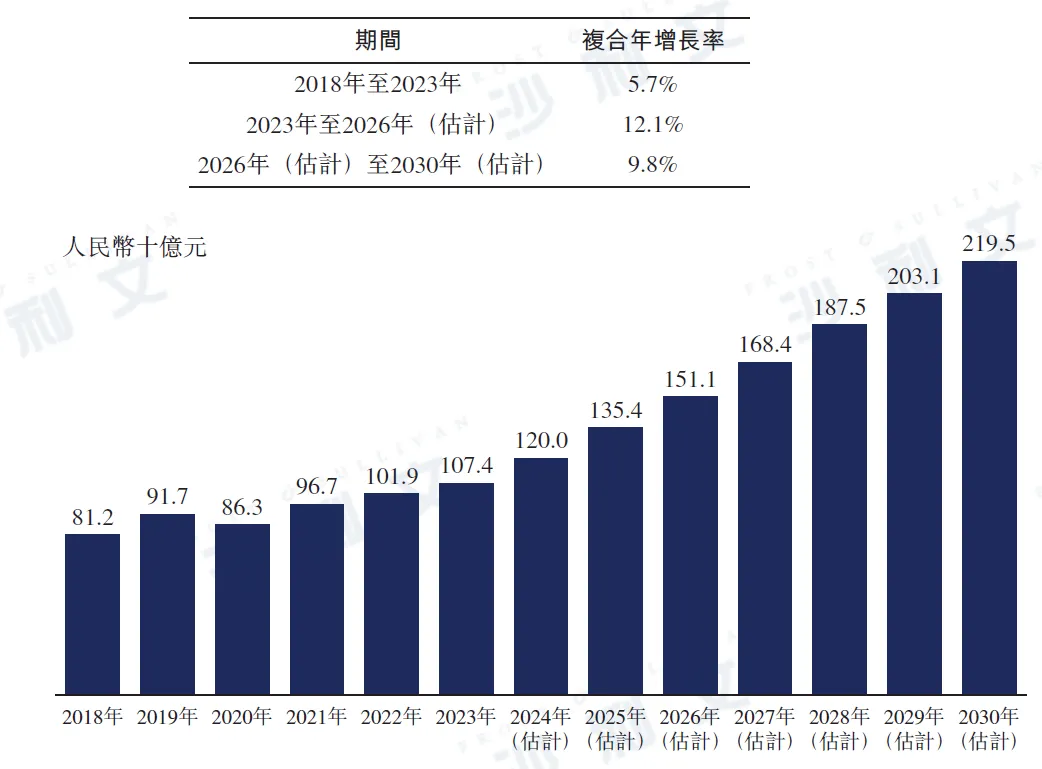
Data source: Analysis by Frost & Sullivan
PART/9
Overview of the Chinese Pulmonary Fibrosis Drug Market
Idiopathic pulmonary fibrosis (IPF) is the most common type of pulmonary fibrosis and is a lung disease that causes scarring (fibrosis) of the lungs. Scarring makes the lungs stiff and leads to breathing difficulties. The lung damage caused by IPF is irreversible and progressive, meaning it will become worse over time. In some cases, specific medications can slow down this gradual deterioration, and sometimes lung transplantation may be recommended for IPF patients. Despite routine treatment, a portion of patients with interstitial lung disease (ILD) develop a progressive phenotype known as fibrotic ILD with progressive phenotype (PF-ILD), characterized by worsening respiratory symptoms, declining lung function, and early death. Population aging, tobacco use, and environmental air pollution have led to a rapid increase in the incidence of IPF. At the same time, the widespread adoption and promotion of medical interventions, as well as education for patients and doctors, will extend the survival time of patients, reduce mortality rates, and increase the base number of IPF patients.
PART/10
Market Size and Forecast of Pulmonary Fibrosis Drugs in China
Pirfenidone and nintedanib (the only two drugs currently available globally for the treatment of IPF) were included in the national medical insurance drug list in 2017 and 2020, respectively, driving rapid growth in the Chinese pulmonary fibrosis drug market from 2018 to 2023. In 2023, the market size of pulmonary fibrosis drugs in China reached approximately RMB 1.5 billion, with a compound annual growth rate of 54.4% from 2018 to 2023. With the continuous increase in the number of approved drugs in the future and the expansion of the indications for these drugs from IPF to other types of pulmonary fibrosis, it is expected that the market size will continue to grow, reaching approximately RMB 2.7 billion by 2026 and RMB 3.2 billion by 2030. The following chart lists the scale of the Chinese pulmonary fibrosis drug market from 2018 to 2030.
China Pulmonary Fibrosis Drug Market, 2018-2030E
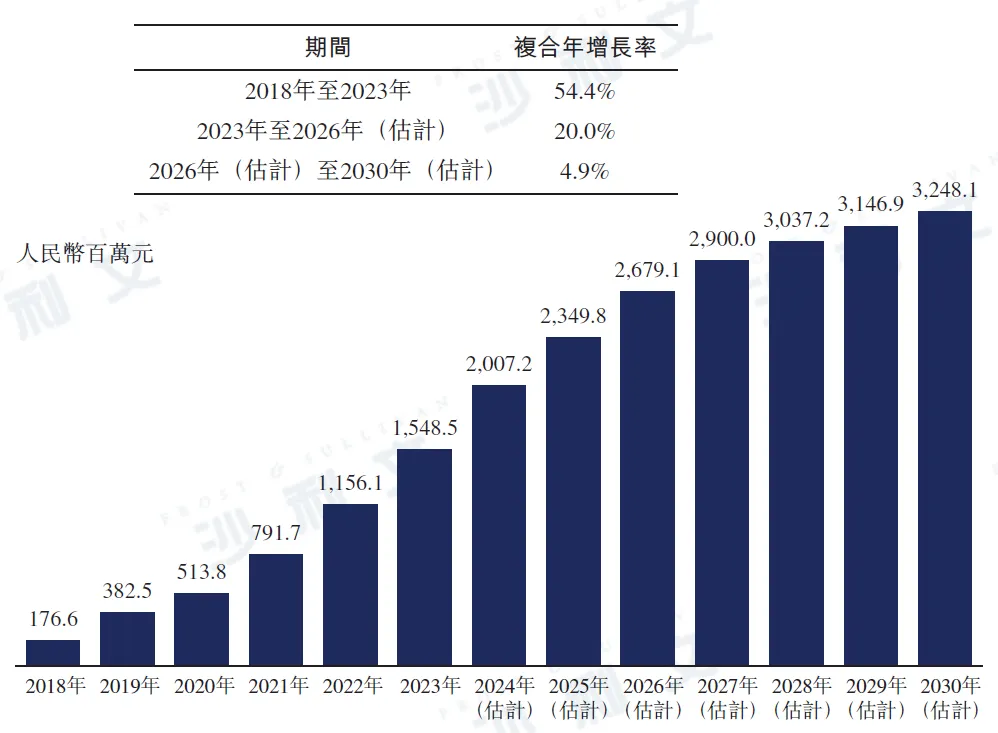
Data source: Analysis by Frost & Sullivan
Click at the end of the articleRead the original textView the full prospectus
Frost & Sullivan, integrating 64 years of global consulting experience, has dedicated 27 years to serving the booming Chinese market. With a global perspective, the firm helps clients accelerate their business growth, achieving industry-leading benchmarks in terms of growth, innovation, and leadership. The healthcare industry is one of the core areas of focus for Frost & Sullivan. Over the past 20-plus years, the Frost & Sullivan team has provided financing and financial advisory services, IPO industry advisory services, strategic consulting, and management consulting to hundreds of outstanding domestic and international biopharmaceuticals, medical devices, healthcare services, and internet healthcare companies. Successful listings include: Weilizhi Bio (9887.HK), Dukang Vision Cloud (2592.HK), Yunzhisheng (9678.HK), TED Medicine (3880.HK), Baise Medical (2609.HK), Yaojie Ankang (2617.HK), Jiangsu Hengrui Medicine (1276.HK), Mire (2629.HK), Ying'en Biotech (9606.HK), Weisheng Pharmaceutical (2561.HK), Yasheng Medicine Group (NASDAQ:AAPG), Brain Motion Aurora (6681.HK), Health Road (2587.HK), Huahao Zhongtian (2563.HK), Yinos (688710.SH), Jingtai Technology (2228.HK), Yimai Sunshine (2522.HK), Shenghe Biotech (2898.HK), Quanxin Biotech (2509.HK), Meizhong Jiahe (2453.HK), WuXi AppTec (2268.HK), Neusoft Xikang (9686.HK), Youzhiyou (2496.HK), Yiming Angke (1541.HK), Corning Biotec (6990.HK), LaiKai Medicine (2105.HK), Lvg Bio (2480.HK), Meis Health (2415.HK), PHECR, Zhongjin Medical (NASDAQ:ZJYL), Meiliyuan (2373.HK), Kangfeng Biotech (6922.HK), Bao'an Biotech (6955.HK), Sididi (1244.HK), Meihao Medical (1947.HK), Gaoshi Medical (2407.HK), Lepu Xin Tai (2291.HK), Jianshi Technology (9877.HK), Health Yuan (JCARE.SW), Lepu Medical (LEPU.SW), Dingdang Health (9886.HK), Bio-Thera (2315.HK), Zhiyun Health (9955.HK), MeinGene (6667.HK), PRE.NASDAQ, Yunkang Group (2325.HK), Ruike Biotech (2179.HK), Lepu Biotech (2157.HK), Baxin Anhuan (2185.HK), Yonghe Medical (2279.HK), Kailai Ying (6821.HK), Beihai Kangcheng (1228.HK), Gusheng Tang (2273.HK), Yingpeng Technology (2251.HK), Minimally Invasive Robotics (2252.HK), Harmony Kaiman (2256.HK), Xianruida (6669.HK), Kangsheng Global (9960.HK), Yimai Tong (2192.HK), Tengsheng Bopai (2137.HK), Canopy (2162.HK), Chaoyu Eye Hospital (2219.HK), Guichuang Tongqiao (2190.HK), Hehuang Medicine (0013.HK), Koji Pharmaceutical (2171.HK), Zhaoke Eye Hospital (6622.HK), Nature Medicine (UPC.NASDAQ), Sainfo Pharmaceutical (6600.HK), Zhaoyan New Drug (6127.HK), Novogene Health (6606.HK), Tianyan Pharmaceutical (ADAG.NASDAQ), Beikang Medical (2170.HK), Jianbimiao Miao (2161.HK), Minimally Invasive Xin Tong (2160.HK), Jiaosisi Pharmaceutical (1167.HK), HepB Pharma (2142.HK), JD Health (6618.HK), Deqi Medicine (6996.HK), Rongchang Biotech (9995.HK), WuXi Biologics (2126.HK), Xiansheng Pharmaceutical (2096.HK), Yunding Newray (1952.HK), Jiahe Biotech (6998.HK), Zai Ding Medicine (9688.HK), Oukang Vision (1477.HK), Yongtai Biotech (6978.HK), Haipure Pharmaceutical (9989.HK), Kuaopei Pharmaceutical (9939.HK), Peijia Medical (9996.HK), Kangfang Biotech (9926.HK), Nuo Cheng Jianhua (9969.HK), Tianjing Biotech (IMAB.NASDAQ), Kanglong Chemical (3759.HK), China Antibody (3681.HK), Dongyao Pharmaceutical (1875.HK), Yasheng Medicine (6855.HK), Fuhong Hanlin (2696.HK), Hansoh Pharmaceutical (3692.HK), Mabotech (2181.HK), Fangda Holdings (1521.HK), Via Biotech (1873.HK), CStone Pharmaceuticals (2616.HK), Junshi Biotech (1877.HK), WuXi Bio-Tech (2359.HK), Xinda Biotech (1801.HK), Hualing Medicine (2552.HK), BeiGene (6160.HK), Galle Pharmaceutical (1672.HK), WuXi Biologics (2269.HK), China Resources Medicine (3320.HK), Yajichen Scientific Research Pharmaceutical (2633.HK), Hualing China Medicine (HCM.NASDAQ), Biotechnology (1548.HK), BBI Life Sciences (1035.HK), Tongyuan Kang Medicine (2410.HK), etc. In terms of the number of filings, the Frost & Sullivan healthcare team maintains an absolute leading position in Hong Kong's healthcare IPO market, consistently ranking first in market share from 2018 to 2023.
Since the listing of the first batch of companies on the Sci-tech Innovation Board in July 2019, Frost & Sullivan reports have been widely cited in the prospectuses of leading listed companies on the Sci-tech Innovation Board, including Hanbang Technology (688755.SH), Zhongyan Shares (688716.SH), Optoglass Technology (688450.SH), Jinghe Integration (688249.SH), Wuxi Rilian (688531.SH), Maolai Optics (688502.SH), Kangwei Century (688426.SH), Jinchuan Protein (688137.SH), Novogene (688428.SH), Aopuima Biotech (688293.SH), MicroPort Electrophysiology (688351.SH), Mengke Pharmaceutical (688373.SH), Yifang Biotech (688382.SH), Jicui Pharmaceutical (688046.SH), Haichuang Pharmaceutical (688302.SH), Rongchang Biotech (688331.SH), Rendu Biotech (688193.SH), Shouyao Holdings (688197.SH), Heyuan Biotech (688238.SH), Yaxin Security (688225.SH), Xidi Micro (688173.SH), Mawei Biotech (688062.SH), Yahong Medicine (688176.SH), BeiGene (688235.SH), Jiameng Medical (688246.SH), Dizhe Medicine (688192.SH), Novozyme (688105.SH), Chengda Biotech (688739.SH), Geke Micro (688728.SH), Huaxi Biotech (688363.SH), Junshi Biotech (688180.SH), Zhejiang Pharma (688266.SH), Beiogen (688177.SH), Shenzhou Cells (688520.SH), etc., are considered one of the most powerful, professional, and influential industry research institutions in the industry. We hope to work with enterprises to understand industry trends, seize development opportunities, jointly promote innovation and upgrading of China's big health industry, and build a healthy future.
Recommended Reads (scroll up and down for more)
Frost & Sullivan assists Veeflex in successfully listing on the Hong Kong Stock Exchange (9887.HK)
Frost & Sullivan helps Dukang Vision Cloud successfully go public in Hong Kong (2592.HK)
Frost & Sullivan helps CloudBee successfully go public in Hong Kong (9678.HK)
Frost & Sullivan helps Mirae achieve successful listing on the Hong Kong Stock Exchange (2629.HK)
Frost & Sullivan helps Health Road successfully go public in Hong Kong (2587.HK)

