Frost & Sullivan
Nanjing Weilizhi Bio-Tech Co., Ltd. (Stock Code: 9887.HK) successfully listed on the main board of the Hong Kong capital market on July 25, 2025. The company is a biopharmaceutical company in the registration clinical stage, aiming to independently discover, develop and commercialize tumor immunotherapies. It focuses on discovering and developing T-cell-centered immunotherapies. Frost & Sullivan (hereinafter referred to as 'Frost & Sullivan') provides exclusive industry advisory services for the listing of Nanjing Weilizhi Bio-Tech Co., Ltd., and hereby warmly congratulates them on their successful listing.

Nanjing Weilizhibo Biotechnology Co., Ltd. (hereinafter referred to as 'Weilizhibo') successfully went public on July 25, 2025. The company plans to issue 3,205.4 million H shares, of which 90% will be international offerings and 10% will be public offerings. The maximum selling price per share is HK$35.00, and the net proceeds from the fundraising are expected to be approximately HK$968 million.
During the process of listing in Hong Kong this time, Frost & Sullivan mainly undertook the following tasks: helping the issuer accurately and objectively understand its positioning in the target market, using objective market data to discover, support, and highlight the issuer's competitive advantages, assisting the issuer, investment banks, and other intermediaries in completing the relevant parts of the prospectus (such as overview, competitive advantages and strategy, industry overview, business, and other important chapters), helping the issuer complete communication with the Hong Kong Stock Exchange and investors, assisting investors in quickly understanding the market ecosystem and competitive landscape, and assisting the issuer in completing feedback on various industry-related issues from the Hong Kong Stock Exchange.
Frost & Sullivan has always been a leader in helping companies go public in Hong Kong. According to LiveReport's big data (statistical data as of June 30, 2025), from January to June 2025, as well as during the past 12 and 36 months, Frost & Sullivan provided listing industry advisory services to 29 (market share 71%), 60 (market share 67%), and 164 (market share 69%) Hong Kong-listed IPOs respectively, ranking first in terms of number. It has rich industry experience and communication skills with regulatory authorities, exchanges, investment and financing institutions, and various related organizations.
PART/1
Investment Highlights
-
The company leverages proprietary excellent bispecific antibodies and other technology platforms to strategically focus on discovering and developing T-cell-centered immunotherapies. The company's target selection is also achieved through research-driven strategies and a comprehensive assessment of regulatory trends and competitive landscape, aiming to fill significant therapeutic gaps and ensure that our products are commercially viable when they enter the market.
-
The company's core product is a PD-L1 and 4-1BB bispecific antibody. It is the world's first therapy targeting the 4-1BB receptor that has entered clinical registration, making 4-1BB an important next immunotherapy target after PD-1/L1, CTLA-4, and LAG3, providing broad opportunities for its treatment of various solid tumors.
-
In multiple clinical trials conducted by the company for extra-pulmonary neuroendocrine carcinoma, small cell lung cancer, cholangiocarcinoma, non-small cell lung cancer, and other solid tumors, LBL-024 has demonstrated encouraging efficacy signals and good safety. In addition, the company received Breakthrough Therapy Designation (BTD) for LBL-024 in the treatment of extra-pulmonary neuroendocrine carcinoma after treatment with core product LBL-024 from the NMPA in October 2024. LBL-024 also received Orphan Drug Designation (ODD) from the FDA in November 2024 for the treatment of neuroendocrine carcinoma.
-
In multiple clinical trials in China, LBL-024 has shown encouraging efficacy signals and good safety profile when used as monotherapy or in combination with chemotherapy for the treatment of advanced extrapulmonary neuroendocrine carcinoma. As of February 12, 2025, the median follow-up period was 18.2 months, and the median overall survival was 11.9 months. The 6-month overall survival rates for the overall population, second-line, and third-line or above populations were 79.5%, 90.0%, and 70.8% respectively. As of February 12, 2025, no dose-limiting toxicity was observed, even at the highest test dose of 15.0 mg/kg, which did not reach the maximum tolerated dose. Most adverse events were grade 1 or 2 and manageable.
-
The company's competitive advantages have attracted strategic partners (such as NewCo established by Aditum Bio) to collaborate with us, enabling us to utilize international clinical resources in a synergistic, efficient, and cost-effective manner, accelerate drug development, and enter overseas markets.
PART/2
Overview of the Oncology Drug Market
In recent years, the Chinese oncology drug market has shown continuous growth, reaching an annual revenue of $382 billion in 2024. The average annual compound growth rate (CAGR) from 2019 to 2024 was 7.6%. It is expected that the market will expand significantly, reaching $574 billion by 2027, with an average annual compound growth rate of 14.5% from 2024 to 2027, and further reaching $800 billion by 2030, with an average annual compound growth rate of 11.7% from 2027 to 2030. Globally, the cancer drug market grew from $143.5 billion in 2019 to $251.4 billion in 2024, with an average annual compound growth rate of 11.9%. This growth is mainly due to the expansion of the patient base and improved access to medical services. It is expected that global oncology market revenue will reach $336.9 billion by 2027, with an average annual compound growth rate of 10.3% from 2024 to 2027, and further reaching $436.8 billion by 2030, with an average annual compound growth rate of 9.0% from 2027 to 2030. The following figure describes the global oncology drug market from 2019 to 2030.

Tumor drugs can be divided into three categories: tumor immunotherapy, chemotherapy, and targeted therapy. Among them, tumor immunotherapy has been growing rapidly, accounting for 26.5% of the tumor drug market in 2023. It is expected that this proportion will continue to expand, reaching 47.9% by 2030. The following chart shows the global tumor market breakdown by therapy from 2023 to 2030.

PART/3
Overview of Tumor Immunotherapy
Immunotherapy is a key area of oncology innovation that leverages the critical role of T cells in the immune system. T cells are essential for the immune system's ability to recognize and destroy cancer cells, and their function is supported and regulated by various signals and interactions within the immune system. Current tumor immunotherapies offer limited clinical benefits mainly due to the complexity and variability of the tumor microenvironment. For example, immune microenvironments lacking T cell infiltration (commonly referred to as 'cold tumors') have a lower objective response rate to PD-1/PD-L1 antibody drugs. Such cold tumors pose significant challenges, including primary and acquired resistance to immunotherapy. Therefore, understanding and addressing the different characteristics of the tumor microenvironment are crucial for improving the therapeutic effectiveness of tumor immunotherapy.
The development of tumor immunotherapy has gone through different stages, each marked by significant progress in treatment strategies. The first wave focused on monoclonal antibody therapy targeting immune checkpoint inhibitors to enhance T cell-mediated anti-tumor responses, achieving an objective response rate of about 20%. Examples include ipilimumab (CTLA-4 inhibitor), pembrolizumab (PD-1 inhibitor), and nivolumab (PD-1 inhibitor). Although PD-1/PD-L1 monotherapy has been proven effective for some patients, it does not generally induce durable responses. Some patients develop resistance or adverse reactions, and the therapy often cannot fully restore the damaged immune response, leading to limited overall effectiveness. The second wave introduced combination therapies by integrating immune checkpoint inhibitors with chemotherapy or anti-angiogenic drugs, significantly improving clinical outcomes and achieving objective response rates of 40% to 50%. Important combinations include nivolumab and ipilimumab in combination for melanoma, as well as pembrolizumab and chemotherapy in combination for non-small cell lung cancer.
The third wave represents a complex approach that systematically targets multiple immune pathways to achieve maximum anti-tumor effects, with the goal of reaching higher objective remission rates. This phase showcases the future trends and mission of tumor immunotherapy, focusing on the comprehensive development of the immune system to combat cancer. In this wave, therapies with emerging targets and broad prospects are emerging, such as antibodies targeting 4-1BB and LAG3. The dual-target strategy against 4-1BB and PD-L1 shows high potential, and the widespread expression of these targets provides significant opportunities for the expansion of highly effective bispecific antibodies in the indications of a range of solid tumors. Similarly, by exploiting the unique mechanism of LAG3 on these frequently co-expressed cells, targeting LAG3 with PD-1 can more effectively activate exhausted T cells than targeting PD-1 alone. However, the characteristics of these antibody tumor drugs are complex structures and high research and development difficulty, reflecting their substantial platform effects. The development process is similar to systems engineering, where selecting the correct target, antibody, and structural design are crucial.
Overview of 4-1BB Antibodies
4-1BB is a member of the TNFR superfamily and an important protein found on the surface of T cells in the immune system. When bound to its ligand, 4-1BBL, which is typically present on antigen-presenting cells, 4-1BB can act as a co-stimulatory molecule, enhancing the activation, survival, and growth of T cells. This interaction enhances the anti-cancer ability of the immune system, making 4-1BB a promising target for next-generation tumor immunotherapy. Therapies targeting 4-1BB are widely recognized due to their broad expression and key role in inducing T cell responses, showing great potential, especially for cancers where PD-1/PD-L1 inhibitors are effective. The effective use of 4-1BB activation can significantly enhance T cell proliferation, demonstrating potential in addressing limitations associated with PD-1 inhibitors. 4-1BB plays a crucial role in the activation and lifespan of T cells, which are key for generating an effective immune response against tumors. Its impact can also be indirectly proven by the success of CAR-T cell therapy, confirming that 4-1BB's involvement can improve efficacy. In addition, 4-1BB is essential for promoting T cell infiltration into and penetration of the tumor microenvironment, which is a key factor for achieving tumor-specific immune responses.
Currently, anti-4-1BB antibody drugs are being developed for the treatment of various cancers, including but not limited to neuroendocrine cancer, small cell lung cancer, non-small cell lung cancer, cholangiocarcinoma, hepatocellular carcinoma, esophageal squamous cell carcinoma, and gastric cancer. These therapies aim to provide more effective, longer-lasting, and safer anti-cancer responses by fully utilizing the body's immune system. In recent years, progress has been made in the development of 4-1BB monoclonal antibodies, and multiple clinical trials are currently underway to evaluate their effectiveness against various cancer types. However, challenges include the toxicity that may arise from overactivation of the immune system, determining the optimal design and dosage for balancing efficacy and side effects, leading to a lack of approved 4-1BB targeted drugs. Bispecific antibodies targeting two molecular pathways are becoming a new strategy. Specifically, bispecific antibodies that target both PD-L1 and 4-1BB have great promise due to their complementary mechanisms of reducing immunosuppression and inducing immune cell activation. This approach can effectively address the limitations of PD-1/PD-L1 monotherapy, which usually has a low remission rate and can lead to resistance. For example, 4-1BB can affect related tumor extracellular processes and change the tumor microenvironment. It is noteworthy that targeting the tumor microenvironment rather than peripheral blood is considered more important for PD-L1 and 4-1BB, especially for tumors with immune rejection or immunoinactivity. Dual targeting of PD-L1 and 4-1BB can not only enable tumor cells to bind and activate dependent T cells to avoid off-target toxicity in the liver but also optimize the participation of anti-tumor immunity.
● Global Competitive Landscape of 4-1BB Antibodies
Globally, multiple participants are developing 4-1BB monoclonal antibodies for solid tumors. Among them, BMS's ublituximab and Pfizer's ustilimab have made significant progress and can serve as examples. Lianzhi Biologics' LVGN6051 is the only candidate product that has entered Phase II trials. The following table lists details of the global 4-1BB monoclonal antibody pipeline:
Main indication markets for MTK inhibitors

The following table details the global pipeline of targeted 4-1BB and PD-L1 bispecific antibodies. LBL-024 has been initiated in July 2024 for a single-arm registration clinical trial in China to treat extra-pulmonary neuroendocrine tumors. As of the latest feasible date, it is the world's first and only immunotherapy targeting 4-1BB that has entered the registration clinical trial phase. In addition to LBL-024, there are currently no other PD-L1/4-1BB bispecific antibodies globally undergoing evaluation through the accelerated approval pathway.

Data source: Analysis by Frost & Sullivan
The following table summarizes the global incidence rates and absolute market sizes for each major indication of LBL-024 during the indicated period:

Data source: Analysis by Frost & Sullivan
The global 4-1BB antibody market is expected to reach $13.2 million, $2.9 billion, and $17.4 billion in 2026, 2030, and 2035, respectively. This is due to the large patient populations for cancer types such as extra-pulmonary neuroendocrine carcinoma, small cell lung cancer, cholangiocarcinoma, esophageal squamous cell carcinoma, non-small cell lung cancer, gastric cancer, melanoma, urothelial carcinoma, and lymphoma (most of which lack targeted therapy). There are over 2 million patients in 2024 and it is expected to exceed 3 million by 2035. This growth reflects an average annual compound growth rate of 284.9% from 2026 to 2030 and 43.1% from 2030 to 2035. This is due to more than 10 global 4-1BB antibody pipelines in clinical trials, and the mechanism can enhance PD-1 remission rates through T cell activation and elevated PD-L1 levels, achieving rapid market penetration and thus accelerating related growth. The US 4-1BB antibody market is expected to reach $13.2 million, $2.3 billion, and $10.2 billion in 2026, 2030, and 2035, respectively, with an average annual compound growth rate of 263.3% from 2026 to 2030 and 34.3% from 2030 to 2035. The Chinese 4-1BB antibody market is expected to reach $900,000, $52.9 million, and $2.8 billion in 2027, 2030, and 2035, respectively, with an average annual compound growth rate of 288.8% from 2027 to 2030 and 121.8% from 2030 to 2035. This growth is supported by the clinical development of alternative treatment options for indications such as extra-pulmonary neuroendocrine carcinoma and small cell lung cancer, which have limited treatment options. The expected average annual compound growth rate from 2027 to 2030 is expected to decline due to a larger base naturally leading to a lower growth rate. The following chart shows the market size of global 4-1BB antibody drugs: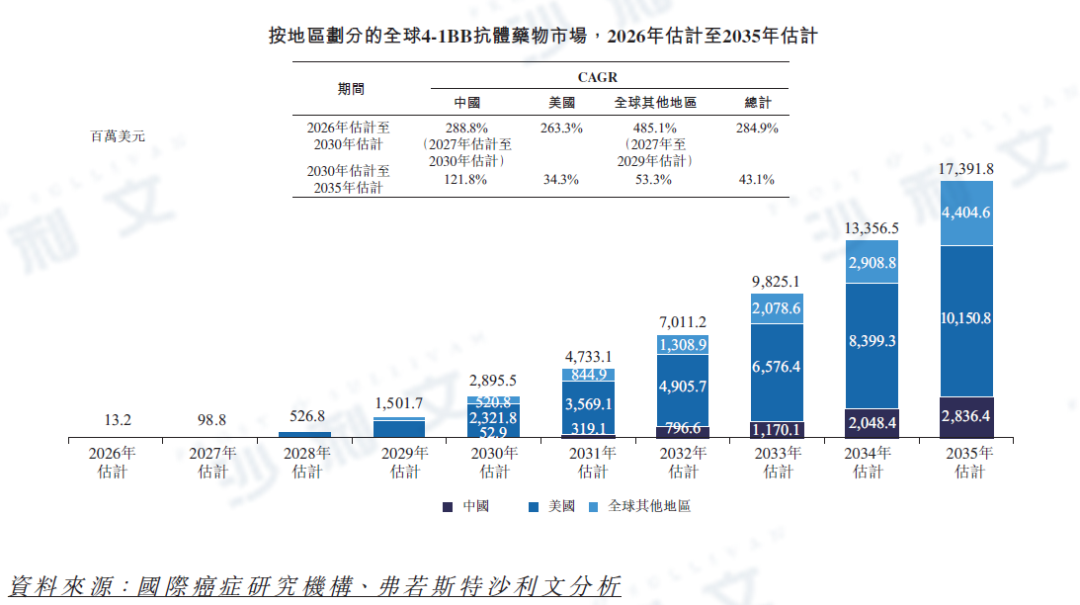
Overview of LAG3 Antibodies
LAG3 is an immune checkpoint receptor on activated T cells, which negatively regulates the activity of these cells through a variety of ligands including MHC-II, LSECtin, Gal-3, and FGL1. Due to its role in antigen presentation, long-term antigen exposure caused by infection or tumors can lead to high and sustained expression of LAG3 on T cells. This results in 'exhaustion' of T cells, loss of their effector function, thereby weakening immune surveillance and allowing tumors to evade the immune system. The mechanism of action of anti-LAG3 antibodies is to block their interaction with ligands by binding to LAG3, inhibit signal pathways, promote T cell proliferation and cytokine secretion, thereby restoring tumor immune surveillance. Currently, antibodies targeting anti-LAG3 are being developed or have been developed for the treatment of various solid tumors.
The use of LAG3 inhibitors in combination with anti-PD-1/PD-L1 drugs has shown synergistic potential. When used together, these therapies can more effectively regulate T cell function, enhance the immune system's ability to recognize and attack tumors. This combination therapy can restore the function of suppressed effector T cells, increase the number of activated CD8+ and CD4+ T cells, and improve their ability to target tumor cells. In addition, preclinical and early clinical studies have shown that compared with PD-1 monotherapy, this combination therapy can improve remission rates and solve a major problem in cancer treatment, namely the development of PD-1 resistance. Global efforts are underway to develop combination therapies involving LAG3 and PD-1/PD-L1 inhibitors. With the FDA's approval of the relatlimab-nivolumab regimen for the treatment of advanced melanoma, this strategy has been clinically validated.
●Global Competitive Landscape of LAG3 Monoclonal Antibodies
In March 2022, the FDA approved the first and only LAG3-targeted antibody combination, Opdualag®, for the treatment of unresectable or metastatic melanoma. In 2023 (the first full year after its commercial launch), Opdualag® achieved global sales of $627 million. As a validated target, LAG3 has attracted the attention of pharmaceutical or biotech companies worldwide and in China. As shown in the following global pipeline table, the drug development pipeline for LAG3 monoclonal antibodies is currently very complete:

Frost & Sullivan, with its 64 years of global consulting experience and 27 years of dedicated service to the booming Chinese market, uses a global perspective to help clients accelerate their business growth, achieving benchmark positions in industry growth, innovation, and leadership. The healthcare industry is one of the core areas of focus for Frost & Sullivan. Over the past 20-plus years, the Frost & Sullivan team has provided services such as financing and financial advisory, IPO industry advisory, strategic consulting, and management consulting to hundreds of outstanding domestic and international biopharmaceutical, medical device, healthcare services, and internet healthcare companies. Successful listings include: Pukang Vision Cloud (2592.HK), Yunzhisheng (9678.HK), TedMed (3880.HK), Baize Medical (2609.HK), Yaojie Ankang (2617.HK), Jiangsu Hengrui Medicine (1276.HK), Mire (2629.HK), Ying'en Biotech (9606.HK), Weisheng Pharmaceutical (2561.HK), Yasheng Pharmaceutical Group (NASDAQ:AAPG), Brainlight (6681.HK), Health Road (2587.HK), Huahao Zhongtian (2563.HK), Yinosi (688710.SH), Jingta Technology (2228.HK), Yimai Sunshine (2522.HK), Shenghe Biotech (2898.HK), Quanxin Biotech (2509.HK), Meizhong Jiahé (2453.HK), WuXi AppTec (2268.HK), Neusoft Xikang (9686.HK), Youzhiyou (2496.HK), Yiming Angke (1541.HK), Corning Biotec (6990.HK), LaiKai Medicine (2105.HK), Lvgreen Biotech (2480.HK), Messe Health (2415.HK), PHECR, Zhongjin Medical (NASDAQ:ZJYL), Meiliyuan (2373.HK), Kangfeng Biotech (6922.HK), Bao'an Biotech (6955.HK), Sidi (1244.HK), Meihao Medical (1947.HK), Gaoshi Medical (2407.HK), Lepu Xintai (2291.HK), Jianshi Technology (9877.HK), Health Yuan (JCARE.SW), Lepu Medical (LEPU.SW), Dingdang Health (9886.HK), Bio-Thera (2315.HK), Zhiyun Health (9955.HK), MeinGene (6667.HK), Prenetics (PRE.NASDAQ), Yunkang Group (2325.HK), Ruike Biotech (2179.HK), Lepu Biotech (2157.HK), Baxin Anhuan (2185.HK), Yonghe Medical (2279.HK), Kailai Ying (6821.HK), Beihai Kangcheng (1228.HK), Gusheng Tang (2273.HK), Yingpeng Technology (2251.HK), Minimally Invasive Robotics (2252.HK), Harmony Kamane (2256.HK), Xianruida (6669.HK), Kangsheng Global (9960.HK), Yimai Tong (2192.HK), Tengsheng Bopharm (2137.HK), Canopy (2162.HK), Chaoyu Eye Hospital (2219.HK), Guichuang Tongqiao (2190.HK), Hehuang Medicine (0013.HK), Koi Pharmaceutical (2171.HK), Zhaoke Eye Hospital (6622.HK), Upc Pharma (UPC.NASDAQ), Saiseng Pharma (6600.HK), Zhaoyan New Drugs (6127.HK), Novogene Health (6606.HK), ADAG Pharma (ADAG.NASDAQ), Beikang Medical (2170.HK), Jianbimiao Miao (2161.HK), Minimally Invasive Heart Tong (2160.HK), Jiaosisi Pharmaceutical (1167.HK), HepB Biotech (2142.HK), JD Health (6618.HK), Deqi Pharma (6996.HK), Rongchang Biotech (9995.HK), WuXi AppTec (2126.HK), SonoGen Biotech (9939.HK), Peijia Medical (9996.HK), Kangfang Biotech (9926.HK), NuoCheng Jianhua (9969.HK), Tianjing Biotech (IMAB.NASDAQ), Kanglong Chemical (3759.HK), China Antibody (3681.HK), Dongyao Pharma (1875.HK), Yasheng Pharmaceutical (6855.HK), Fuhong Hanlin (2696.HK), Hansoh Pharmaceutical (3692.HK), Mabot Biotech (2181.HK), Fangda Holdings (1521.HK), Via Bio (1873.HK), CStone Pharmaceuticals (2616.HK), Junshi Biosciences (1877.HK), WuXi AppTec (2359.HK), Xinda Biotech (1801.HK), Hailong Medicine (2552.HK), BeiGene (6160.HK), Gilead Sciences (1672.HK), WuXi AppTec (2269.HK), China Resources Medicine (3320.HK), Yager Scientific Research Pharmaceutical (2633.HK), HCM China Medicine (HCM.NASDAQ), Biotech (1548.HK), BBI Life Sciences (1035.HK), Tongyuan Kang Medicine (2410.HK), etc. In terms of the number of listed projects, the Frost & Sullivan healthcare team maintains an absolute leading position in Hong Kong healthcare IPOs, consistently ranking first in market share from 2018 to 2023.
自2019年7月科创板首批公司挂牌以来,沙利文报告也被广泛引用于业内领先的科创板上市公司的招股文件中,包括:汉邦科技(688755.SH)、中研股份(688716.SH)、光格科技(688450.SH)、晶合集成(688249.SH)、无锡日联(688531.SH)、茂莱光学(688502.SH)、康为世纪(688426.SH)、近岸蛋白(688137.SH)、诺诚健华(688428.SH)、奥浦迈生物(688293.SH)、微创电生理(688351.SH)、盟科药业(688373.SH)、益方生物(688382.SH)、集萃药康(688046.SH)、海创药业(688302.SH)、荣昌生物(688331.SH)、仁度生物(688193.SH)、首药控股(688197.SH)、和元生物(688238.SH)、亚信安全(688225.SH)、希荻微(688173.SH)、迈威生物(688062.SH)、亚虹医药(688176.SH)、百济神州(688235.SH)、嘉和美康(688246.SH)、迪哲医药(688192.SH)、诺唯赞(688105.SH)、成大生物(688739.SH)、格科微(688728.SH)、华熙生物(688363.SH)、君实生物(688180.SH)、泽璟制药(688266.SH)、百奥泰(688177.SH)、神州细胞(688520.SH)等,被认为是业内最具实力、最具专业度、最具影响力的行业研究机构之一。我们希望与企业一起,洞悉行业前瞻,抓住发展契机,共同推动中国大健康产业的创新和升级,铸就健康未来。
推荐阅读
沙利文助力亚盛医药集团成功赴美上市(NASDAQ:AAPG)
沙利文助力药华药成功发行GDR并在卢森堡证券交易所上市(PHECR)
沙利文助力健康元成功发行GDR并在瑞交所上市(JCARE.SW)
沙利文助力乐普医疗成功发行GDR并在瑞交所上市(LEPU.SW)
*以上顺序不分先后,按上市时间倒序排列

