Sichuan Kerun Biopharmaceutical Co., Ltd. (Stock Code: 6990.HK) successfully listed on the Hong Kong capital market on July 11, 2023. The company is a comprehensive and integrated innovative biopharmaceutical company dedicated to the research, development, manufacturing, and commercialization of innovative drugs. It can strategically and rapidly advance 33 differentiated pipelines, of which 13 are in clinical stages. The company has accumulated over a decade of experience in ADC development and recently reached an agreement with Merck & Co. to develop up to seven preclinical ADC assets, which is the largest biopharmaceutical licensing transaction received by a Chinese company to date. Frost & Sullivan (hereinafter referred to as 'Frost & Sullivan') provides exclusive industry advisory services for the listing of Sichuan Kerun Biopharmaceutical Co., Ltd., and hereby warmly congratulate them on their successful listing.

Sichuan Kerunbo Tai Biopharmaceutical Co., Ltd. (hereinafter referred to as 'Kerunbo Tai') successfully listed on July 11, 2023, with a global issuance of 2,244.61 million shares at a price of HK$60.60 per share, raising approximately HK$1.2589 billion.
During the Hong Kong listing process, Frost & Sullivan mainly undertook the following tasks: helping the issuer accurately and objectively understand its positioning in the target market, using objective market data to discover, support, and highlight the issuer's competitive advantages, assisting the issuer, sponsor, and other professional intermediary institutions in completing the writing of relevant parts of the prospectus (such as the overview, competitive advantages and strategy, industry overview, business, and other important sections), assisting the issuer in communicating with the Hong Kong Stock Exchange and investors, helping investors quickly understand the market ecosystem and competitive landscape, and assisting the issuer in completing feedback on industry-related issues from the Hong Kong Stock Exchange.
Investment highlights
The company is one of the first in China and among the few globally to establish an integrated ADC platform OptiDC;
The company has multi-platform technical expertise and has built a comprehensive anti-tumor drug pipeline;
The company has established an integrated drug development capability that encompasses R&D, production, quality control, and commercialization;
The company has an experienced leadership team;
According to the Frost & Sullivan report, as of 2022, the company:
It is the first Chinese company to license internally discovered and developed ADC candidate drugs to the top ten global biopharmaceutical companies;
The collaboration with Merck Daxcel to develop up to seven preclinical ADC assets is the largest biopharmaceutical licensing transaction received by a Chinese company to date;
A140 is the first phase 3 head-to-head trial design of a cetuximab biosimilar in China, which strictly follows the 'Guidelines for the Design of Biologics Clinical Trials of Cetuximab Injection (Trial Implementation)' issued by the Center for Drug Evaluation.
SKB336 is China's first domestically developed FXI/FXIa drug that has entered clinical trials.
Overview of the Antibody Drug Market in China
The antibody drug market encompasses antibodies used for tumor and non-tumor indications. Antibody drugs are the largest category of biologics, typically more effective than traditional chemical drugs (such as chemotherapy) with fewer side effects, as they aim to utilize specific molecular targets involved in the disease's pathogenesis and may reduce harm to non-target cells. The main categories of antibody drugs include monoclonal antibodies (mAbs), antibody-drug conjugates (ADCs), and bispecific antibodies.
As of the last practicable date, monoclonal antibodies (mAbs) are the largest category of antibody drugs in China, with a market size reaching 742 billion yuan in 2022. New-generation antibody drugs such as ADCs and bispecific antibodies have great therapeutic potential. With more candidate drugs being approved, it is expected that the ADC and bispecific antibody markets will grow significantly in the near future, at a rate faster than the overall antibody drug market.
Data source: Analysis by Frost & Sullivan
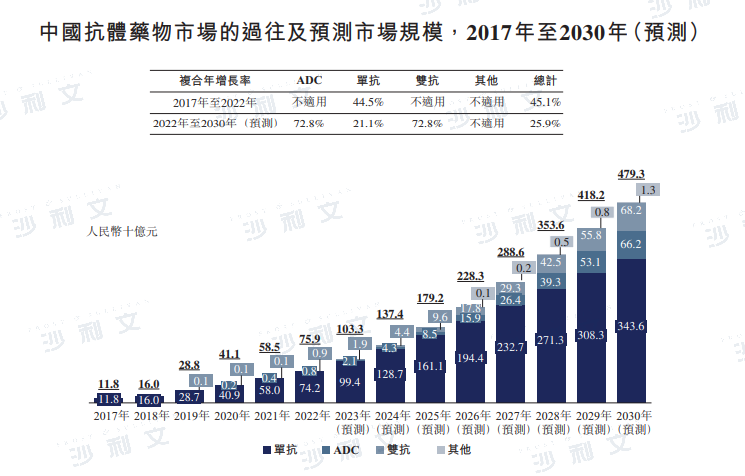
Overview of the Scale of Chinese ADCs
The global market size of ADCs has rapidly grown from $1.6 billion in 2017 to $7.9 billion in 2022, with a compound annual growth rate of 37.3%. It is expected to continue growing at a compound annual growth rate of 30.0% from 2022 to 2030. After the National Medical Products Administration approved the first ADC drug, Herceptin, in 2020, the Chinese ADC market began to grow, with an expected increase from RMB 800 million in 2022 to RMB 662 billion, a compound annual growth rate of 72.8%.
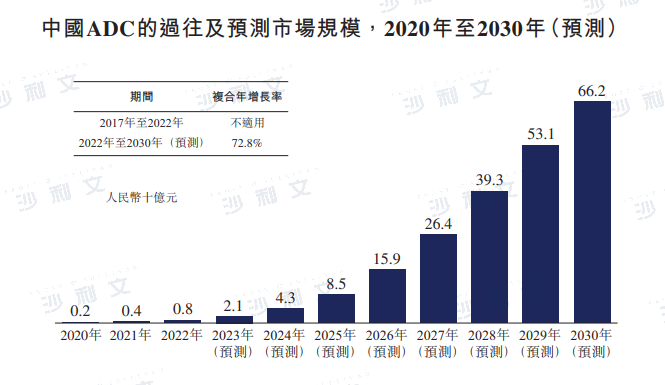
Data source: Analysis by Frost & Sullivan
Market Drivers and Future Trends for ADC Development
1) Progress in ADC design and coupling technology.After the first FDA approval in 2000, ADCs have shown a recent development momentum following twenty years of repeated trials. In particular, current research on ADC technology and cancer biology is expected to drive the discovery of new molecular targets and payloads, as well as better linker designs and conjugation techniques. This could lead to new designs that improve the therapeutic effects of ADCs and reduce various toxic issues limiting the use of existing commercially available ADCs.
2) Expand indications and treatment lines.With the continuous progress in ADC design and conjugation technology, a total of 12 ADCs have been approved by the FDA to date. ADCs have evolved from a treatment option for relapsed and refractory hematological malignancies to a promising front-line treatment paradigm, applicable to a wider range of solid tumor indications and other indications. The advancement of ADC technology is expected to expand the potential molecular targets and indications, including non-tumor diseases such as autoimmune diseases. ADCs are also expected to enter earlier treatment lines, extending to the treatment of early-stage cancers and benefiting more patient populations.
3) Combine with other treatment modalities.The mechanism of action of ADCs can synergize with other treatment modalities to enhance the killing power against tumor cells. For example, combined therapies that combine ADCs with immun checkpoint inhibitors hold great promise for enhancing anti-tumor efficacy in clinical studies.
4) The demand for comprehensive integration capabilities is increasing day by day.The R&D of ADCs requires extensive proprietary technologies and capabilities in biology, chemistry, and production, covering areas ranging from biologics to small molecules and bio-processing. The development and production of ADCs are in increasing demand, which is expected to benefit biopharmaceutical companies with comprehensive end-to-end integration capabilities. Such capabilities will enable ADC candidate drugs to develop rapidly.
Global and China's TROP2 ADC Market Overview
TROP2 is a transmembrane protein that plays a significant role in embryonic and organ development, with low expression in normal tissues. In a wide range of cancers, TROP2 is often overexpressed and promotes cancer proliferation, invasion, and metastasis.
The global market for TROP2 ADCs was $700 million in 2022, and it is expected to grow at a compound annual growth rate of 57.6% starting from 2022, reaching $259 billion by 2030. After the National Medical Products Administration approved Trodelvy, the first TROP2 ADC in China, the market for TROP2 ADCs in China is expected to grow, with an expected compound annual growth rate of 103.0% starting from 2023, reaching RMB 236 billion by 2030.
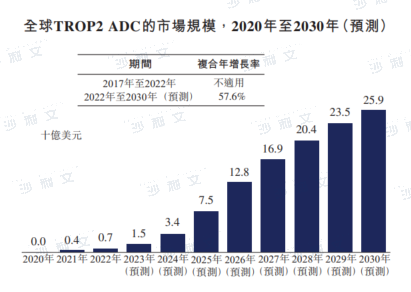
Data source: Analysis by Frost & Sullivan
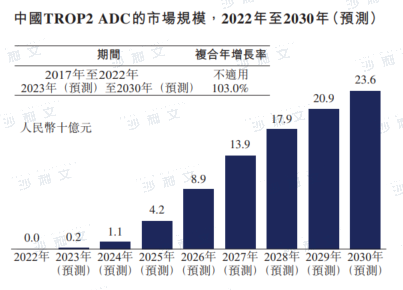
Data source: Analysis by Frost & Sullivan
Competitive landscape of the global TROP2 ADC market
As at the latest practicable date, Gillette Science's Trodelvy®It is the only TROP2 ADC approved in the United States for advanced TNBC, advanced UC, and HR+/HER2-BC, and it is also the only TROP2 ADC approved by the National Medical Products Administration for the treatment of advanced TNBC. As of the same date, there are three TROP2 ADC candidate drugs globally in phase 2 or above, including SKB264 and Trodelvy.®(The drug code is IMMU-132) and DS-1062. The following table shows the global competitive landscape of TROP2 ADCs:

Data source: Analysis by Frost & Sullivan
In the biopharmaceutical industry, Frost & Sullivan has served companies such as BeiGene, Rongchang Biotech, Hutchison MediTech, Fosun Pharma, and Kangfang Biotech.

Click at the end of the articleRead the original textView the full prospectus of Colombo Bay
Frost & Sullivan, with a global consulting experience spanning 62 years and dedicated service to the booming Chinese market for 25 years, helps clients accelerate their business growth with a global perspective, achieving industry-leading benchmarks in growth, innovation, and leadership. The health industry is one of Frost & Sullivan's core areas of focus. Over the past 20-plus years, the Frost & Sullivan team has provided financing financial advisory, IPO industry advisory, strategic consulting, management consulting, and other services to hundreds of outstanding domestic and international biopharmaceuticals, medical devices, healthcare services, and internet healthcare companies. Successful listings include: LaiKai Medicine (2105.HK), Kedil Group (2487.HK), Green Bamboo Biotech (2480.HK), Meis Health (2415.HK), PHECR, Zhongjin Medical (NASDAQ:ZJYL), Yisheng Biotech (NASDAQ:YS), Meiliyuan (2373.HK), Kangfeng Biotech (6922.HK), Bo'an Biotech (6955.HK), Sididi (1244.HK), Meihao Medical (1947.HK), Gaoshi Medical (2407.HK), Lepu Xin Tai (2291.HK), Jian Shi Technology (9877.HK), Health Yuan (JCARE.SW), Lepu Medical (LEPU.SW), Dingdang Health (9886.HK), Bio-Thera (2315.HK), Zhiyun Health (9955.HK), MeinGene (6667.HK), Prenetics (PRE.NASDAQ), Yunkang Group (2325.HK), Rike Biotech (2179.HK), Lepu Biotech (2157.HK), Clear Medical (1406.HK), Baxin An (2185.HK), Yonghe Medical (2279.HK), Kailai Ying (6821.HK), Beihai Kangcheng (1228.HK), Gusheng Tang (2273.HK), Yingtong Technology (2251.HK), Clover Biotech (2197.HK), Minimally Invasive Robotics (2252.HK), Harmony Kamman (2256.HK), Kunbo Medical (2216.HK), Xianruida (6669.HK), Kangsheng Global (9960.HK), Yimai Tong (2192.HK), Tengsheng Bopharm (2137.HK), Canopy (2162.HK), Chaoyu Eye Hospital (2219.HK), Guichuang Tongqiao (2190.HK), Huihuang Medicine (0013.HK), Koi Pharmaceutical (2171.HK), Zhaoke Eye Hospital (6622.HK), Nature Medicine (UPC.NASDAQ), Saiseng Pharmaceutical (6600.HK), Zhaoyan New Drugs (6127.HK), Novogene Health (6606.HK), Tianyan Pharmaceutical (ADAG.NASDAQ), Beikang Medical (2170.HK), Jianbimiao Miao Miao (2161.HK), Minimally Invasive Xin Tong (2160.HK), Rui Li Medical Beauty (2135.HK), Jiake Pharmaceutical (1167.HK), HepB Pharma (2142.HK), JD Health (6618.HK), Deqi Medicine (6996.HK), Rongchang Biotech (9995.HK), WuXi AppTec (2126.HK), SinoBIO (2096.HK), Yunding New Energy (1952.HK), Jiahe Biotech (6998.HK), Zai Ding Medicine (9688.HK), Ocular Biotech (1477.HK), Yongtai Biotech (6978.HK), Haipu Pharmaceutical (9989.HK), Kechuang Pharmaceutical (9939.HK), Peijia Medical (9996.HK), Kangfang Biotech (9926.HK), Nuo Cheng Jian Hua (9969.HK), Tianjing Biotech (IMAB.NASDAQ), Kanglong Chemical (3759.HK), China Antibody (3681.HK), Dongyao Pharmaceutical (1875.HK), Yasheng Medicine (6855.HK), Fuhong Hanlin (2696.HK), Hansoh Pharmaceutical (3692.HK), Mabtech (2181.HK), Fangda Holdings (1521.HK), Via Biotech (1873.HK), Cornerstone Pharmaceuticals (2616.HK), Junshi Biotech (1877.HK), WuXi AppTec (2359.HK), Innovent Biologics (1801.HK), Hailun Medicine (2552.HK), BeiGene (6160.HK), Gilead Sciences (1672.HK), WuXi AppTec (2269.HK), China Resources Medicine (3320.HK), Yakuten Scientific Research Pharmaceutical (2633.HK), Huihuang China Medicine (HCM.NASDAQ), Kingsbridge Biotechnology (1548.HK), BBI Life Sciences (1035.HK), etc. In terms of the number of filings, the Frost & Sullivan healthcare team maintains an absolute leading position in Hong Kong healthcare IPOs, consistently ranking first in market share from 2018 to 2022.
Since the listing of the first batch of companies on the Sci-tech Innovation Board in July 2019, Frost & Sullivan reports have also been widely cited in the prospectuses of leading listed companies on the Sci-tech Innovation Board, including: Jinghe Integration (688249.SH), Wuxi Rilian (688531.SH), Maolai Optics (688502.SH), Kangwei Century (688426.SH), Jinqian Protein (688137.SH), Nuo Cheng Jianhua (688428.SH), Aopu Mai Biotech (688293.SH), MicroPort Electrodiagnostics (688351.SH), Mengke Pharmaceutical (688373.SH), Yifang Biotech (688382.SH), Jicui Yaokang (688046.SH), Haichuang Pharmaceutical (688302.SH), Rongchang Biotech (688331.SH), Rendu Biotech (688193.SH), Shouyao Holdings (688197.SH), Heyuan Biotech (688238.SH), Yaxin Security (688225.SH), Xidi Micro (688173.SH), Mawei Biotech (688062.SH), Yahong Medicine (688176.SH), BeiGene (688235.SH), Jiamhe Meikang (688246.SH), Dizhe Medicine (688192.SH), Novogene (688105.SH), Chengda Biotech (688739.SH), Geke Micro (688728.SH), Huaxi Biotech (688363.SH), Junshi Biotech (688180.SH), Zhejiang Oncology (688266.SH), BeiGene (688177.SH), Shenzhou Cells (688520.SH), etc. They are considered one of the most powerful, professional, and influential industry research institutions in the sector. We hope to work with enterprises to understand industry trends, seize development opportunities, jointly promote innovation and upgrading of China's health industry, and build a healthy future.
Recommended Reading
Frost & Sullivan assists Lüzhoubio in successfully listing on the Hong Kong Stock Exchange (2480.HK)
Frost & Sullivan helps MesHealth successfully go public in Hong Kong (2415.HK)
Frost & Sullivan assists Zhongjin Medical in successfully going public in the US (NASDAQ: ZJYL)
Frost & Sullivan assisted Yisheng Biology in successfully going public on the NASDAQ (NASDAQ:YS)
Frost & Sullivan assists Meiliyuan in successfully listing on the Hong Kong Stock Exchange (2373.HK)
Frost & Sullivan helps Thundersoft successfully go public in Hong Kong (1244.HK)
Frost & Sullivan helps Lepuventec successfully go public in Hong Kong (2291.HK)
Frost & Sullivan helps Zhiyun Health successfully go public in Hong Kong (9955.HK)
Frost & Sullivan assists MeinGene in successfully listing on the Hong Kong Stock Exchange (6667.HK)
Frost & Sullivan assisted Prenetics in successfully going public on the NASDAQ (NASDAQ:PRE)
Frost & Sullivan assists Clear Medical in successfully going public in Hong Kong (1406.HK)
Frost & Sullivan assists Kaleido in successfully listing on the Hong Kong Stock Exchange (6821.HK)
Frost & Sullivan helps Gushengtang successfully go public in Hong Kong (2273.HK)
Frost & Sullivan assisted Yuhuan in successfully listing on the Hong Kong Stock Exchange (2256.HK)
Frost & Sullivan assists Kunbo Medical in successfully going public in Hong Kong (2216.HK)
Frost & Sullivan assists Conocoalt in successfully listing on the Hong Kong Stock Exchange (2162.HK)
Frost & Sullivan assists Nature Pharmaceuticals in successfully going public on the NASDAQ:UPC)
Frost & Sullivan helps Jianbaimiao Miao successfully go public in Hong Kong (2161.HK)
Frost & Sullivan helps JD Health successfully go public in Hong Kong (6618.HK)
Frost & Sullivan helps develop PharmaCure successfully go public in Hong Kong (9939.HK)
Frost & Sullivan assisted Tianjing BIO in successfully going public on the NASDAQ (NASDAQ: IMAB)
Frost & Sullivan assists ViaBio in successfully listing on the Hong Kong Stock Exchange (1873.HK)
Frost & Sullivan helps Cornerstone Pharmaceuticals successfully go public in Hong Kong (2616.HK)
Frost & Sullivan helps WuXi AppTec successfully go public in Hong Kong (2359.HK)
Frost & Sullivan helps BeiGene successfully go public in Hong Kong (6160.HK)
Frost & Sullivan helps WuXi AppTec Biologics successfully go public in Hong Kong (2269.HK)
*The above order is not sequential and is arranged in reverse chronological order based on listing time.

