
Jiangsu Ruiko Biotechnology Co., Ltd. (hereinafter referred to as 'Ruiko Biotech') successfully listed on March 31, 2022, with a global issuance of 30,854,500 shares at a price of HK$24.80 per share.
During the Hong Kong listing process, Frost & Sullivan mainly undertook the following tasks: helping the company accurately and objectively understand its positioning in the target market, using objective market data to discover, support, and highlight the company's competitive advantages, assisting the company, investment banks, and other intermediaries in completing relevant parts of the prospectus (such as the overview, competitive advantages and strategy, industry overview, business, and other important sections), facilitating communication with the Hong Kong Stock Exchange and investors, helping investors quickly understand the market ecosystem and competitive landscape, and assisting the company in completing feedback on various industry-related issues from the Hong Kong Stock Exchange.
Vaccine Market Overview
Global vaccine market
The global vaccine market occupies an important position in the global pharmaceutical market. In terms of sales revenue, the global vaccine market size grew from $275 billion in 2016 to $399 billion in 2020, accounting for 3.1% of the total global pharmaceutical market. The compound annual growth rate from 2016 to 2020 was 9.7%. Driven by the emergence of innovative vaccines and the growth of developing country markets, it is expected that the global vaccine market will reach $1292 billion by 2030, with a compound annual growth rate of 12.5% from 2020 to 2030.
Global vaccine market size, 2016 - 2030E
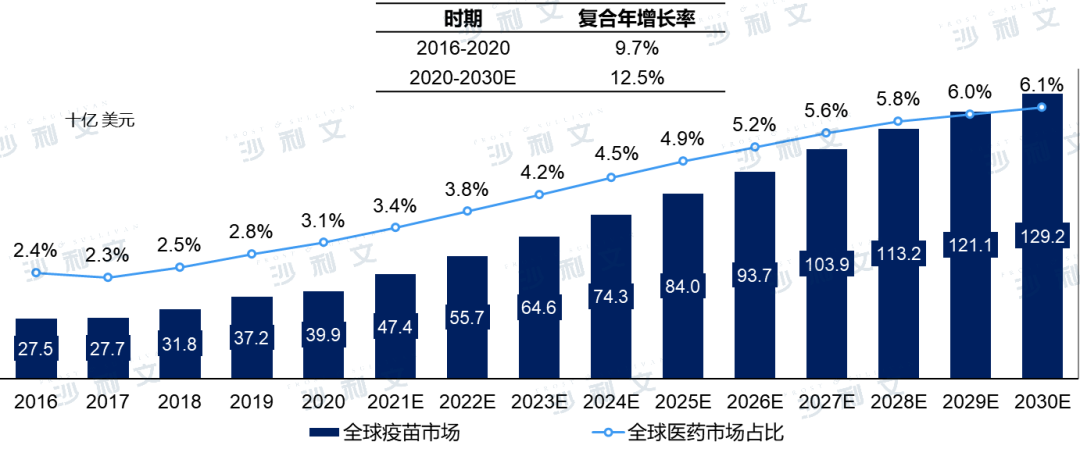
Note: The COVID-19 vaccine market is not currently considered.
Source: Frost & Sullivan report
China vaccine market
In 2020, China was the world's second-largest vaccine market, accounting for more than one-fifth of the global vaccine market. Driven by the increasing popularization of innovative vaccines, favorable government policies, continuous technological innovation in vaccines, and growing awareness of vaccination, the output value of China's vaccine market has grown from 271 billion yuan in 2016 to 753 billion yuan in 2020, with a compound annual growth rate of 29.1%. It is expected to reach 3333 billion yuan by 2030, with a compound annual growth rate of 16.0% from 2020 to 2030.
China Vaccine Market Output Value, 2016-2030E
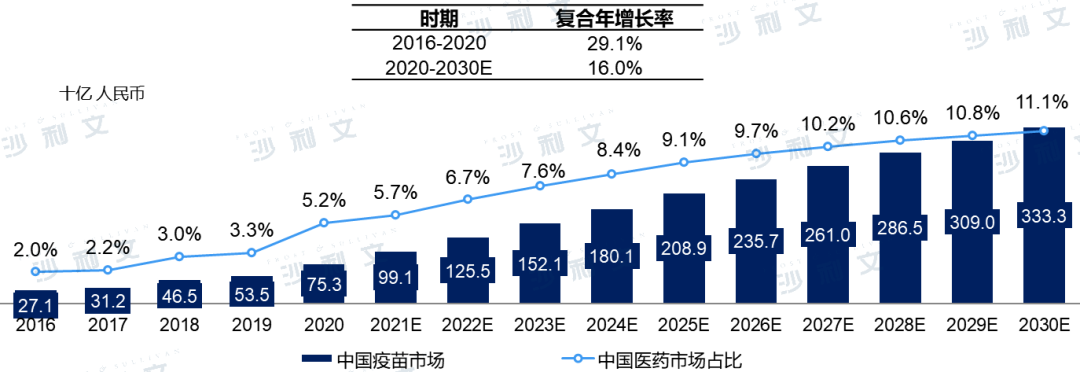
Note: The market scale in China is based on the output value predicted using 2020 batch issuance data. The COVID-19 vaccine market is not currently considered. Historical data comes from the National Institutes for Food and Drug Control (NIFDC), which is highly regulated and depends on policies set by the government and the efficiency of NIFDC's work that year. The forecast data is based on the assumption that future efficiency of NIFDC will not experience significant fluctuations, while the market continues to be driven by the current unmet demand in China's vaccine industry.
Source: Frost & Sullivan report
Vaccines based on whole pathogens are considered traditional vaccines, including inactivated vaccines, attenuated live vaccines, and split vaccines. Other types of technical approaches, such as polysaccharide vaccines, conjugate vaccines, toxoid vaccines, peptide vaccines, virus-like particle vaccines, recombinant protein vaccines, nucleic acid vaccines, and viral vector vaccines, are all considered subunit vaccines. In recent years, the market share of subunit vaccines in China has significantly increased. In 2016, subunit vaccines accounted for 34.7% of the output value of China's vaccine market. With the development of vaccine technology and the introduction of subunit vaccines, by 2020, this proportion had increased to 55.4%, and it is expected that by 2030, subunit vaccines will account for 86.6% of the output value of China's vaccine market.
Value of China's vaccine market, by technology route
2016-2030E
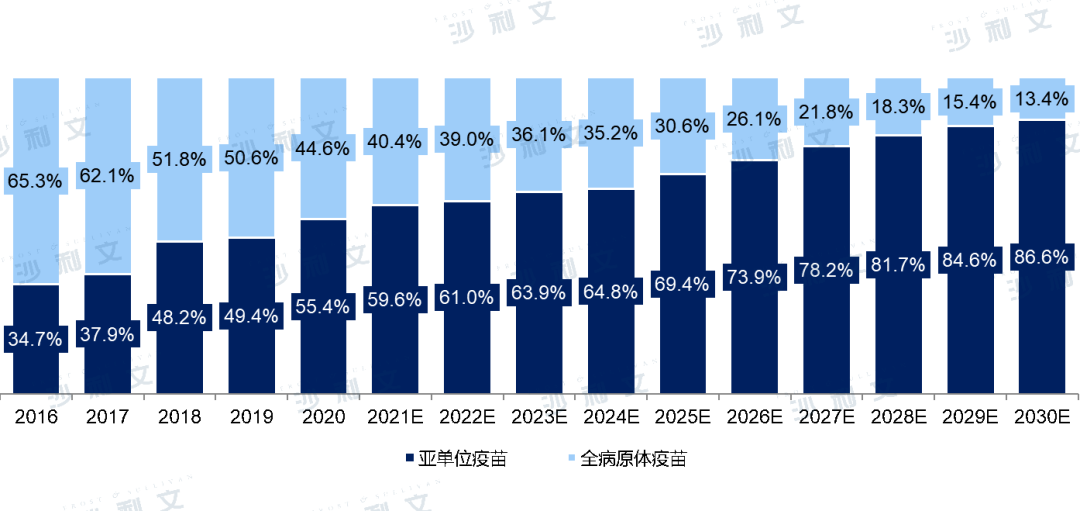
Note: The COVID-19 vaccine market is not currently considered.
Source: Frost & Sullivan report
Adjuvant Overview
Adjuvants are substances that assist in antigenic reactions and stimulate or suppress immune responses. The main functions of adjuvants include: enhancing the immunogenicity of vaccines, altering the nature of immune responses, reducing the amount of antigen required, and decreasing the number of vaccinations needed. To date, only five new adjuvants have been used in vaccines approved by the FDA, including AS04, AS03, MF-59, AS01, and CpG 1018.
Human Papillomavirus Vaccine Market
Overview of Human Papillomavirus
Human papillomavirus (HPV) is the most common pathogen in the reproductive tract. Although most HPV infections can resolve on their own within a few months without any intervention, some infections persist and develop into cervical cancer. Moreover, all HPV serotypes that can infect women can also infect men and cause serious diseases in men, including penile cancer, anal cancer, head and neck cancer, etc. These high-risk HPV infections are mainly caused by HPV types 16, 18, 31, 33, 45, 52, and 58, accounting for about 90% of cervical cancer cases globally, with HPV types 16 and 18 accounting for about 70% of global cervical cancer cases. In addition, HPV types 6 and 11 cause about 90% of anal and genital warts globally. In 2020, cervical cancer caused 4,290 deaths in the United States and 59,060 deaths in China.
Value of the human papillomavirus vaccine market
In China, the HPV vaccine market has enormous potential and is expected to grow rapidly in the coming years. In 2017, China approved the first HPV vaccine, and since then, the market value of HPV vaccines reached 135 billion yuan in 2020, with an estimated 690 billion yuan by 2030. The compound annual growth rate from 2020 to 2030 is 17.7%. By 2030, the market for the 9-valent HPV vaccine is expected to occupy the largest share of the HPV vaccine market. In addition, the market for the 2-valent HPV vaccine is expected to grow rapidly because 2-valent vaccines are usually cheaper and more accessible to those with limited affordability.
China's HPV vaccine market output value, 2016-2030E
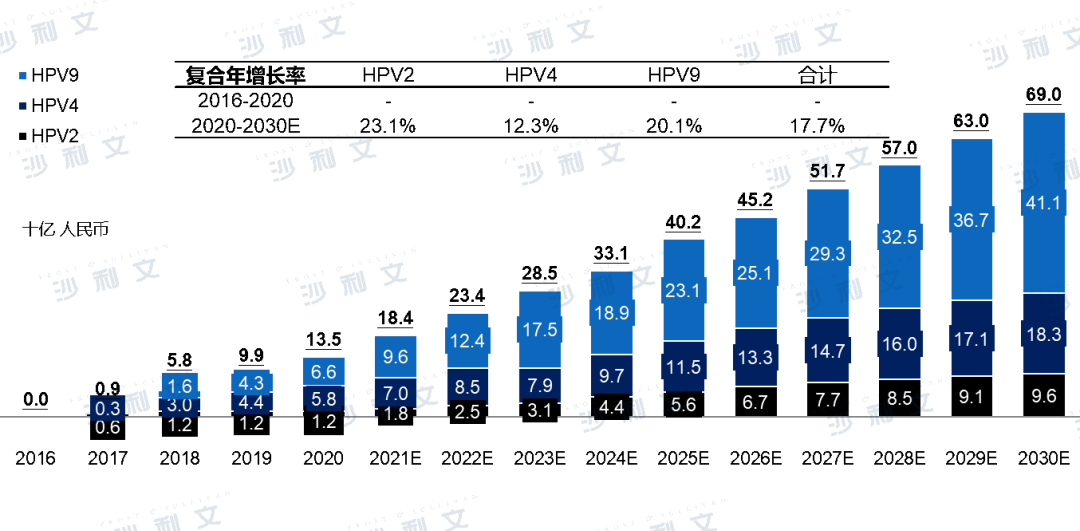
Note: The market size is based on output value and does not include combined vaccines. During the forecast period, no new non-immunization program vaccines were considered for inclusion in the immunization program. Historical data comes from the National Institutes for Food and Drug Control (NIFDC) of China, which are highly regulated and depend on policies formulated by the government and the efficiency of NIFDC's work that year. Forecast data is based on the assumption that there will be no significant fluctuations in the future efficiency of NIFDC's work, while the market continues to be driven by the current unmet demand in China's vaccine industry.
Source: Frost & Sullivan report
COVID-19 vaccine market
COVID-19 is a global pandemic caused by SARS-CoV-2 infection. Since the end of 2019, the COVID-19 pandemic has brought devastating social and economic impacts to China and the world. According to the World Health Organization report, as of March 11, 2022, there have been more than 452 million confirmed cases of COVID-19 globally, claiming more than 6 million lives, and the virus is still spreading worldwide. More than 10.7 billion doses of COVID-19 vaccines have been administered globally.
Supported by the global actions launched by the World Health Organization and governments around the world, COVID-19 vaccines are being rapidly developed. There are five technical routes for COVID-19 vaccines, including recombinant protein vaccines, inactivated vaccines, viral vector vaccines, mRNA vaccines, and DNA vaccines. As of March 11, 2022, a total of 179 COVID-19 candidate vaccines are in clinical development worldwide, including 61 recombinant protein vaccines; 32 COVID-19 vaccines have been approved, including 11 recombinant protein vaccines; and 67 candidate vaccines are in phase III or later stages. Among all commercialized or clinical trials of COVID-19 recombinant protein vaccines, ReCOV is the only vaccine that uses specific combinations of NTD and RBD as antigens.
Herpes Zoster Vaccine Market
Overview of Herpes Zoster
Herpes zoster is a disease caused by the reactivation of varicella-zoster virus (VZV) latent in the sensory ganglia of the posterior root ganglia of the spinal cord or cranial nerves when the body is stimulated or resistance declines. Herpes zoster can occur at any age, but it is most common in the elderly. VZV can be transmitted from people with active herpes zoster to those who have never had chickenpox or been vaccinated. After recovery from chickenpox, VZV can remain dormant in the posterior root ganglia and cranial nerves for decades.
Herpes Zoster Vaccine Market Output Value
Driven by the continuous improvement in people's understanding of herpes zoster and the increasing availability of herpes zoster vaccine products, the market value of herpes zoster vaccines in China is expected to grow significantly from 2.6 billion yuan in 2020 to 29.8 billion yuan in 2025, and further to 562 billion yuan by 2030. The compound annual growth rate from 2020 to 2030 is expected to be 35.8%.
China herpes zoster vaccine market output value, 2016-2030E
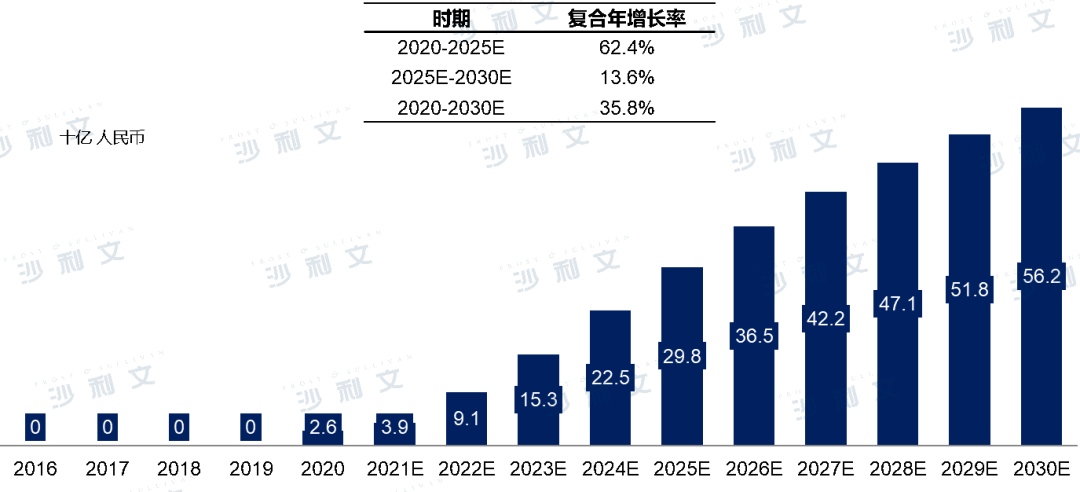
Source: Frost & Sullivan report
Adult Tuberculosis Vaccine Market
Tuberculosis is a disease caused by Mycobacterium tuberculosis (M. tb), which is a major health problem worldwide. Multidrug-resistant tuberculosis (MDR-TB) is a type of tuberculosis caused by strains that are highly resistant to both isoniazid and rifampicin (the two most commonly used anti-tuberculosis drugs), showing resistance to other anti-tuberculosis drugs, or having no resistance at all. MDR-TB has become a global issue. According to data from the World Health Organization, tuberculosis ranked first among global deaths from infectious diseases and parasitic diseases in 2019. In 2019, there were 10 million new active tuberculosis cases globally, nearly 90% of which were adults. China is one of the 30 high-burden countries listed by the World Health Organization for tuberculosis, with the third highest number of new tuberculosis cases in 2019, with a total incidence rate of 775,800 cases.
To date, China has only one adult tuberculosis vaccine, Vaccae®, which was approved in June 2021 for the prevention of pulmonary tuberculosis in people with latent Mycobacterium tuberculosis infection. Another candidate vaccine for adult tuberculosis is undergoing clinical trials in China.
Influenza vaccine market
Influenza Overview
Influenza is an infectious respiratory disease caused by the influenza virus, which can infect the nose, throat, and sometimes the lungs. Influenza can lead to varying degrees of illness, and sometimes even death. The best way to prevent influenza is to get vaccinated against it every year. People over 65 years old, children under 5 years old, and some patients with chronic diseases have a relatively higher risk of developing severe complications if they are infected with influenza. According to data from the National Center for Disease Control and Prevention, in 2020, there were approximately 1.1 million new influenza cases and 70 deaths in China.
Influenza vaccine market output value
In 2017, the National Medical Products Administration approved the first quadrivalent influenza vaccine. Since then, the market value of quadrivalent influenza vaccines in China increased to 4.3 billion yuan in 2020 and is expected to reach 28 billion yuan by 2030, with a compound annual growth rate of 20.7% from 2020 to 2030.
Value of China's Quadrivalent Influenza Vaccine Market, 2016-2030E
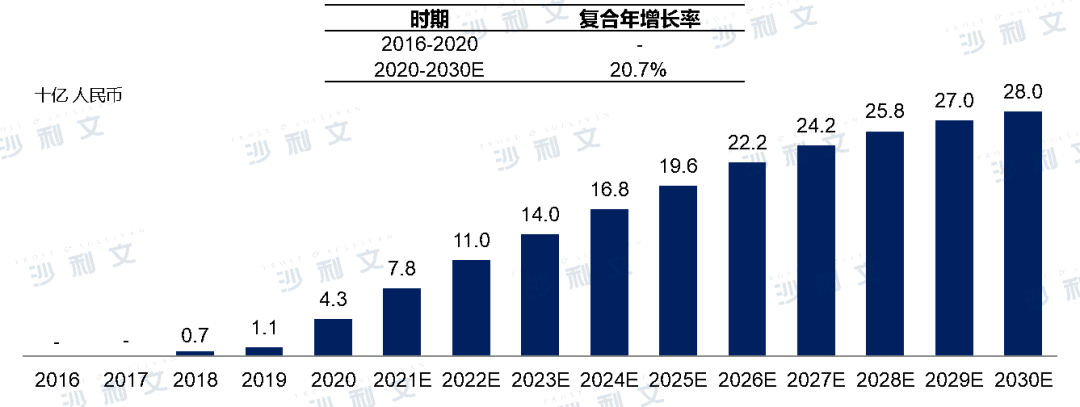
Note: The market size is based on output value and does not include combined vaccines. During the forecast period, no new non-immunization program vaccines were considered for inclusion in the immunization program. Historical data comes from the National Institutes for Food and Drug Control (NIFDC) of China, which is highly regulated and depends on policies formulated by the government and the efficiency of NIFDC's work in that year. Forecast data are based on the assumption that there will be no significant fluctuations in the future efficiency of NIFDC, while the market continues to be driven by the current unsatisfied demand in China's vaccine industry.
Source: Frost & Sullivan report
Hand, foot and mouth disease vaccine market
Hand, foot and mouth disease is a mild infectious disease caused by viral infection. It is more common in children aged 5 and under, accompanied by oral ulcers and rashes on the hands and feet. In 2020, the incidence of hand, foot and mouth disease ranked fourth among legally reportable infectious diseases in China, with more than 760,000 reported cases.
Common viruses causing hand, foot, and mouth disease include Enterovirus 71 (EV71), Coxsackievirus A16 (CA16), Coxsackievirus A10 (CA10), and Coxsackievirus A6 (CA6). These viruses account for nearly 90% of hand, foot, and mouth disease cases in China. Currently, the inactivated EV71 vaccine is the only approved vaccine for hand, foot, and mouth disease globally and in China. In China, EV71 only causes 44% of hand, foot, and mouth disease cases. In 2020, the market value of the inactivated EV71 vaccine in China reached 2.7 billion yuan, with an expected growth to reach 4.6 billion yuan by 2030.
Frost & Sullivan, integrating 61 years of global consulting experience, has dedicated 24 years to serving the booming Chinese market. With a global perspective, we help clients accelerate their business growth and achieve benchmark positions in industry growth, innovation, and leadership. The healthcare industry is one of Frost & Sullivan's core areas of focus. Over the past seventeen years, the Frost & Sullivan healthcare team has provided financing and financial advisory services, IPO industry advice, strategic consulting, management consulting, and other services to hundreds of outstanding domestic and international biopharmaceuticals, medical devices, healthcare services, and internet healthcare companies. Successful listings include: Leopar Biotech (2157.HK), Clear Medical (1406.HK), Bexian An (2185.HK), Yonghe Medical (2279.HK), Kailaiying (6821.HK), Beihai Kangcheng (1228.HK), Gushengtang (2273.HK), Yingpeng Technology (2251.HK), Clover Biotech (2197.HK), Minimally Invasive Robotics (2252.HK), Huarun Kaiman (2256.HK), Kunbo Medical (2216.HK), Xianruida (6669.HK), Kangsheng Global (9960.HK), Yimaitong (2192.HK), Tengsheng Bopai (2137.HK), CanSino Biologics (2162.HK), Chaoyu Ophthalmology (2219.HK), Guichuang Tongqiao (2190.HK), Huarong Medicine (0013.HK), Kexi Pharmaceutical (2171.HK), Zhaoke Ophthalmology (6622.HK), Nature Pharmaceuticals (UPC.NASDAQ), Sainbio (6600.HK), Zhaoyan New Drugs (6127.HK), Novogene Health (6606.HK), ADAG.NASDAQ, Beikang Medical (2170.HK), Jianbimiao Miao Miao (2161.HK), Minimally Invasive Heart Link (2160.HK), Ruili Medical Beauty (2135.HK), Jiake Pharmaceutical (1167.HK), Hepcon Pharma (2142.HK), JD Health (6618.HK), Deqi Pharma (6996.HK), Rongchang Biotech (9995.HK), WuXi AppTec (2126.HK), SinoBIO (2096.HK), Yunding New Energy (1952.HK), Jiahe Biotech (6998.HK), Zai Ding Pharma (9688.HK), Ocular Implant Sciences (1477.HK), Yongtai Biotech (6978.HK), Hapu Pharma (9989.HK), Kepro Pharma (9939.HK), Peijia Medical (9996.HK), Kangfang Biotech (9926.HK), Nuo Cheng Jian Hua (9969.HK), Tian Jing Biotech (IMAB.NASDAQ), Kanglong Chemical (3759.HK), China Antibody (3681.HK), Dongyao Pharmaceutical (1875.HK), Yasheng Pharmaceutical (6855.HK), Fuhong Hanlin (2696.HK), Hansoh Pharmaceutical (3692.HK), Mabtech (2181.HK), Fangda Holdings (1521.HK), Via Biotech (1873.HK), CStone Pharmaceuticals (2616.HK), Junshi Biosciences (1877.HK), WuXi AppTec (2359.HK), Xinda Biosciences (1801.HK), Hualing Medicine (2552.HK), BeiGene (6160.HK), Gilead Sciences (1672.HK), WuXi Biologics (2269.HK), China Resources Medicine (3320.HK), Yacogen Scientific Research Pharmaceutical (2633.HK), Huarong China Medicine (HCM.NASDAQ), Gencell Biotechnology (1548.HK), BBI Life Sciences (1035.HK), etc. In terms of the number of listed projects, the Frost & Sullivan healthcare team maintains an absolute leading position in Hong Kong's healthcare IPO market, consistently ranking first in market share from 2018 to 2021.
Since the listing of the first batch of companies on the Sci-tech Innovation Board in July 2019, Frost & Sullivan reports have been widely cited in the prospectuses of leading Sci-tech Innovation Board-listed companies in the industry, including: CapitalBio (688197.SH), Heyuan Biotech (688238.SH), Yaxin Security (688225.SH), Xidi Microelectronics (688173.SH), Mawei Biotechnology (688062.SH), Yahong Medicine (688176.SH), BeiGene (688235.SH), Jiahe Meikang (688246.SH), Dizhe Medicine (688192.SH), Novogene (688105.SH), Chengda Biology (688739.SH), Geke Microelectronics (688728.SH), Huaxi Biosciences (688363.SH), Junshi Biosciences (688180.SH), Zhejiang Genomics (688266.SH), BeiGene (688177.SH), and Shenzhou Cells (688520.SH). They are considered to be the most powerful, professional, and influential industry research institutions in the sector. We hope to work with enterprises to understand industry trends, seize development opportunities, jointly promote innovation and upgrading of China's healthcare industry, and build a healthy future.
Recommended Reading
Frost & Sullivan helps Saisun Pharma successfully go public in Hong Kong (6600.HK)
40. Frost & Sullivan assists Zai Lab to successfully list on the Hong Kong Stock Exchange (9688.HK)
48. Frost & Sullivan assisted Tianjing Biology in successfully going public in the US (IMAB.NASDAQ)

