Cutia Therapeutics (Stock Code: 2487.HK) successfully listed on the Hong Kong capital market on June 12, 2023. Cutia Therapeutics is a research and development biopharmaceutical company focused on dermatology, with an emphasis on the broad field of dermatological treatment and care, including drugs for local fat accumulation management, hair diseases and care, skin diseases and care, and epidermal anesthesia. The company has established a pipeline comprising one core product and eight candidate drugs, among which the core product CU-20401 is a recombinant mutant collagenase targeting obesity, overweight, and other metabolic diseases related to local fat accumulation. Frost & Sullivan (hereinafter referred to as 'Frost & Sullivan') provides exclusive industry advisory services for Cutia Therapeutics' listing, and we hereby extend our warmest congratulations on its successful listing.

Kedex Group successfully went public on June 12, 2023, with a global offering of 2,128.18 million shares, including 212.84 million shares sold in Hong Kong and 1,915.34 million shares sold internationally. The issue price was HK$21.85, raising a net amount of approximately HK$392.7 million.
During the Hong Kong listing process, Frost & Sullivan mainly undertook the following tasks: helping the issuer accurately and objectively understand its positioning in the target market, using objective market data to discover, support, and highlight the issuer's competitive advantages, assisting the issuer, sponsor, and other professional intermediary institutions in completing the writing of relevant parts of the prospectus (such as the overview, competitive advantages and strategy, industry overview, business, and other important sections), facilitating communication between the issuer and the Stock Exchange and investors, helping investors quickly understand the market ecosystem and competitive landscape, and assisting the issuer in completing feedback on industry-related issues from the Stock Exchange.
Investment highlights
The company focuses on the broad field of dermatological treatment and care, which has great market potential and unmet needs;
The company has a multi-mechanism, highly collaborative drug pipeline;
The company possesses comprehensive capabilities covering the entire broad spectrum of dermatological treatment and care industries;
Relying on its own CATAME® technology platform, the company has continuous R&D and innovation capabilities;
The company has a management team with rich industry experience and foresight;
According to the Frost & Sullivan report, as of 2022, the company:
Possesses the only recombinant mutant collagenase drug in clinical development for the management of localized adiposity in China;
It owns the world's first approved topical finasteride product for androgenetic alopecia and is the only one in China that is in clinical trials.
Overview of the Chinese Market for Local Fat Accumulation Management Drugs
A person's weight can be in three states: normal weight, overweight, and obesity. Overweight and obesity are defined as abnormal or excessive fat accumulation that increases the risk of non-communicable diseases. Subcutaneous fat accumulation is a typical manifestation of obesity and overweight. Currently, available anti-obesity treatment methods on the market include systemic and local treatments. Systemic treatment reduces fat accumulation by regulating endocrine or metabolism through oral or subcutaneous administration of drugs such as orlistat or GLP-1 receptor agonists, rather than reducing local fat accumulation in specific areas. Local treatment reduces local fat accumulation through the use of energy-deficit diets, local fat management drugs, and liposuction surgery. Local fat management drug therapy is applied to the subcutaneous layer to prevent excessive subcutaneous fat, which is a minimally invasive treatment in dermatology with less postoperative pain. These treatments can be applied to the subcutaneous fat within the body, including but not limited to the submental area and abdomen. Some local fat management drugs can further treat obesity, overweight, or other metabolic diseases related to local fat accumulation.
The market for local fat management drugs in China is expected to increase from RMB 8.67 billion in 2023 to RMB 51.44 billion in 2025, with a compound annual growth rate of 143.6% from 2023 to 2025. By 2030, the market for local fat management drugs in China is expected to reach RMB 24.399 billion.
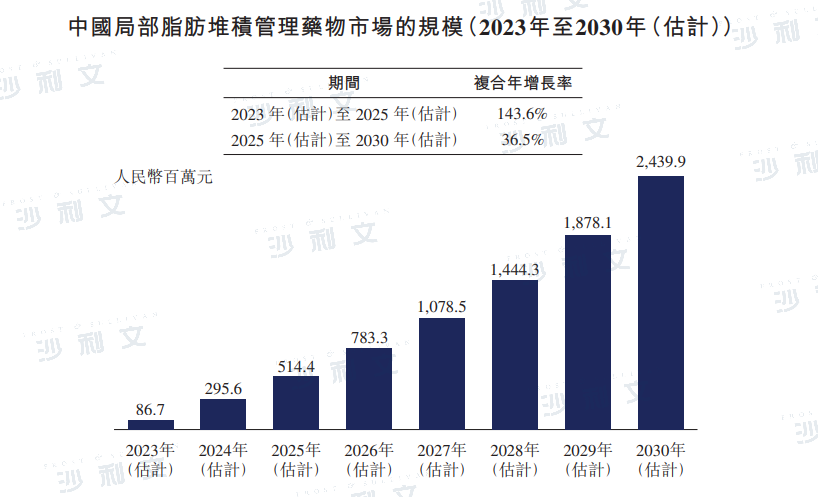
Data source: Analysis by Frost & Sullivan
As of the latest practicable date, China does not have any approved drugs for local fat management. Three candidate products are in clinical trial stages in China, including two deoxycholic acid drugs and CU-20401. CU-20401 is a recombinant mutant collagenase that can degrade extracellular matrix collagen in the subcutaneous fat layer, induce apoptosis of fat cells, effectively reduce fat accumulation, and has mild catalytic activity, reducing the side effects of wild-type collagenase. CU-20401 has completed Phase I clinical trials in human subjects for the treatment of submental fat accumulation (submental fat) and is currently conducting another Phase I clinical trial for the treatment of abdominal fat accumulation (abdominal fat).
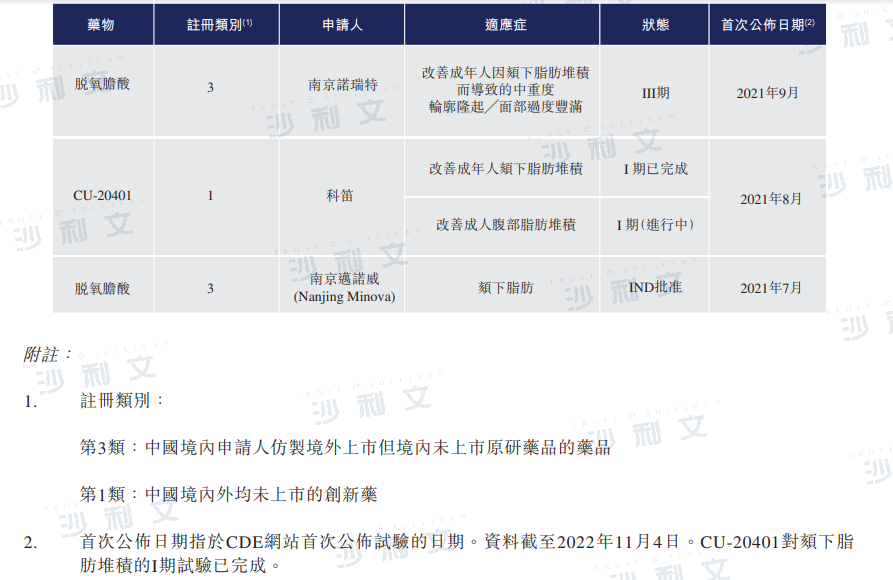
Source: CDE, Frost & Sullivan analysis
Overview of the Hair Disease and Care Market in China
Hair loss is a common hair disorder that can be divided into congenital and acquired hair loss. Acquired hair loss includes non-scarring and scarring hair loss, with the latter leading to permanent hair loss. Androgenetic alopecia and alopecia areata are two of the most common types of hair loss, both belonging to the category of non-scarring hair loss.
The market for hair loss treatment and care products in China increased from RMB 762 billion in 2017 to RMB 1069 billion in 2021, with a compound annual growth rate of 8.8%. It is expected that by 2025, the market will reach RMB 1443 billion, with a compound annual growth rate of 7.8% from 2021 to 2025. By 2030, the market for hair loss treatment and care products in China is expected to reach RMB 2035 billion.
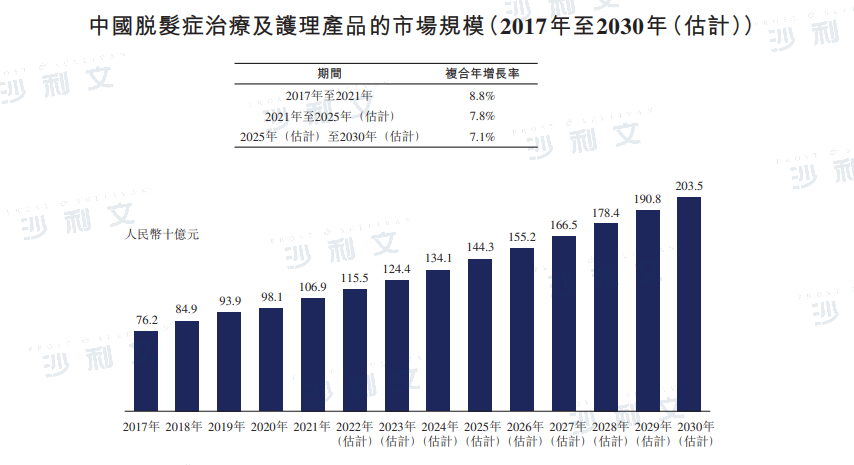
Data source: Analysis by Frost & Sullivan
The drug treatment for androgenetic alopecia mainly comes in two dosage forms: topical medications and oral medications. In China, there is currently one approved drug for the treatment of male androgenetic alopecia for each dosage form: finasteride (oral medication) and minoxidil (topical medication). Topical medications are more readily accepted by patients due to their relatively lower risk of side effects. The table below shows the competitive landscape of approved drugs for androgenetic alopecia in China:
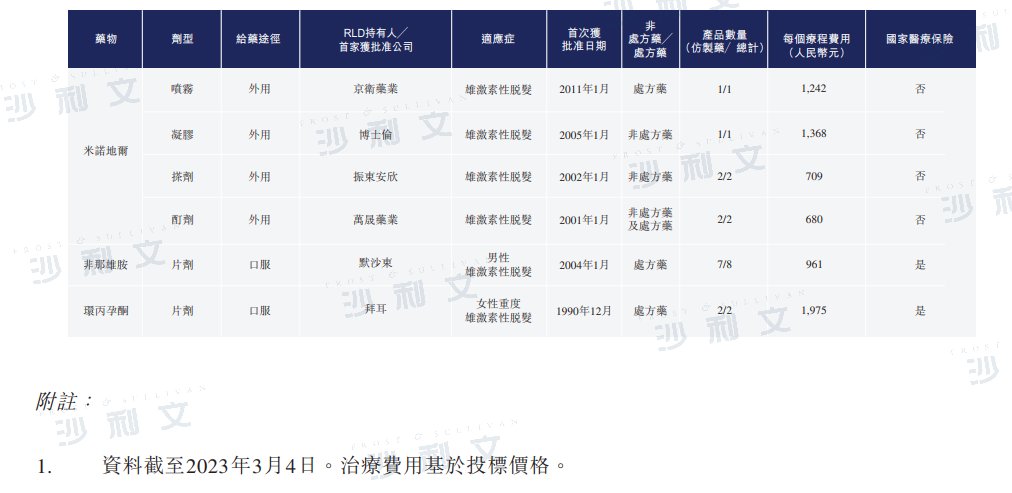
Source: NMPA, analysis by Frost & Sullivan
CU-40102 is the only topical finasteride product in clinical development for androgenetic alopecia in China. The table below shows the competitive landscape of androgenetic alopecia drugs undergoing clinical trials in China:
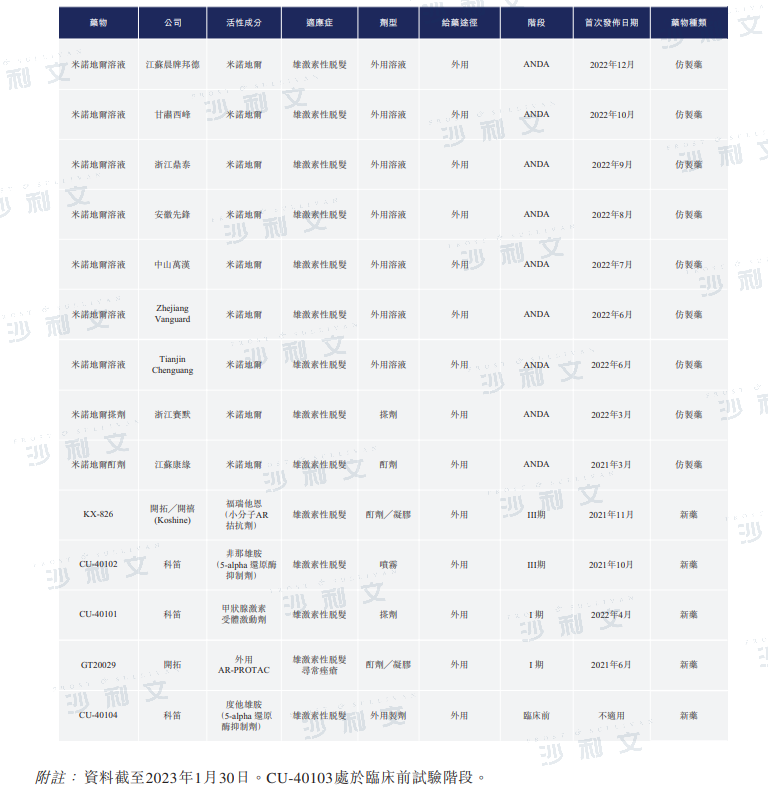
Source: CDE, Frost & Sullivan analysis
Overview of the Chinese Skin Disease and Nursing Market
Skin diseases mainly originate from functional disorders, infections, allergies, or genetic abnormalities of the skin and its appendages, such as common acne, atopic dermatitis, psoriasis, etc. Skin diseases affect the quality of life of patients physiologically, socially, and psychologically.
The Chinese skin disease and care market increased from RMB 218.7 billion in 2017 to RMB 352.6 billion in 2021, with a compound annual growth rate of 12.7%. It is expected that by 2025, the market will reach RMB 493.3 billion, with a compound annual growth rate of 8.8% from 2021 to 2025. By 2030, the Chinese skin disease and care market is expected to reach RMB 740.2 billion.
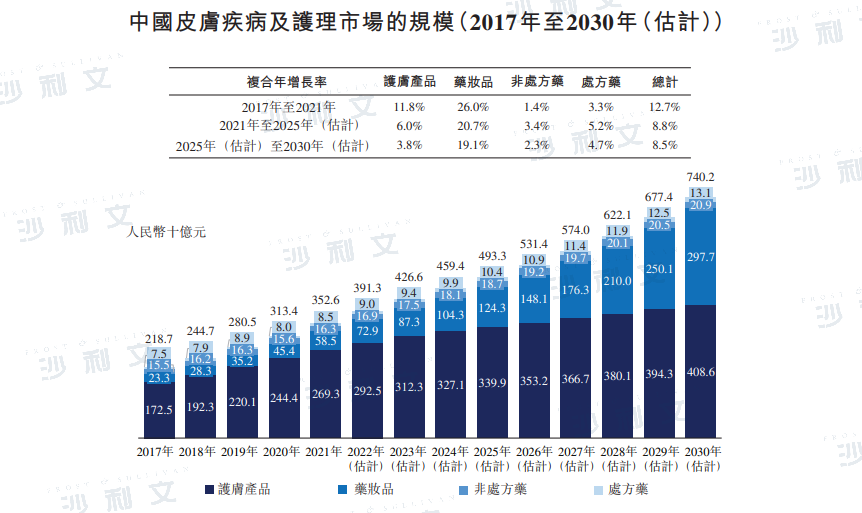
Data source: Analysis by Frost & Sullivan
Overview of the Epidermal Anesthesia Market in China
Epidermal anesthetics are highly penetrating epidermal numbing agents that are sprayed or applied to the skin, mucous membranes, conjunctiva, and other surfaces to render them insensitive to pain. Epidermal anesthetics can be used in consumer and clinical settings. In consumer scenarios, they are typically applied before superficial skin surgical procedures. In clinical practice, they are used before puncture procedures and superficial tissue surgeries. Additionally, they can serve as a pre-treatment for infiltration anesthesia combined with other anesthetics.
The market for epidermal anesthesia products in China increased from RMB 5.04 billion in 2017 to RMB 9.86 billion in 2021, with a compound annual growth rate of 18.3%. It is expected to reach RMB 17.414 billion by 2025, with a compound annual growth rate of 15.3% from 2021 to 2025. By 2030, the market for epidermal anesthesia products in China is expected to reach RMB 26.904 billion.
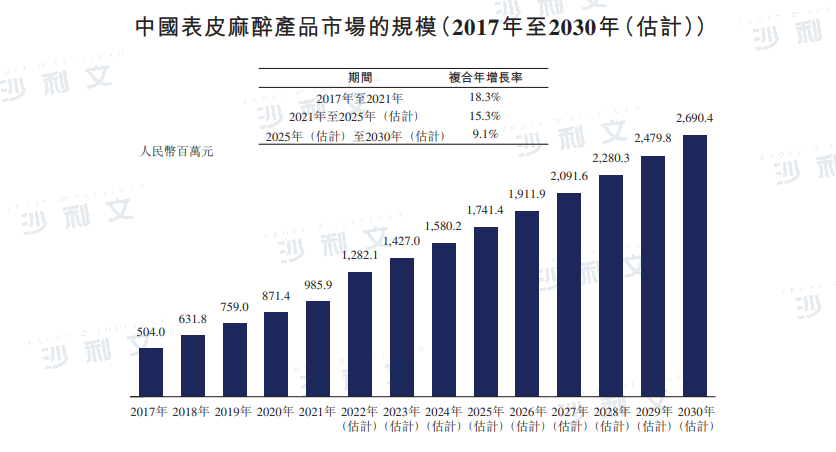
Data source: Analysis by Frost & Sullivan
In the skin care sector, Frost & Sullivan has served companies such as Kexora Pharma, CanSino Biologics, Sany Pharmaceutical, Kangfang Biotech, Mabotech, Myoview Biotechnology, and JuZi Biotechnology.

Click at the end of the articleRead the original textView the full prospectus of Keddi Group
Frost & Sullivan, with a global consulting experience spanning 62 years and a dedicated 25-year service to the booming Chinese market, helps clients accelerate their corporate growth through a global perspective, achieving industry growth, innovation, and leading benchmarks. The healthcare industry is one of Frost & Sullivan's core areas of focus. Over the past 20-plus years, the Frost & Sullivan team has provided financing and financial advisory, IPO industry advisory, strategic consulting, management consulting, and other services to hundreds of outstanding domestic and international biopharmaceuticals, medical devices, healthcare services, and internet healthcare companies. Successful listings include: Laurus Biotech (2480.HK), Meis Health (2415.HK), PHECR, Zhongjin Medical (NASDAQ:ZJYL), Yisheng Biotech (NASDAQ:YS), Meiliyuan (2373.HK), Kangfeng Biotech (6922.HK), Bo'an Biotech (6955.HK), Sidi (1244.HK), Meihao Medical (1947.HK), Gaosheng Medical (2407.HK), Lepu Xin Tai (2291.HK), Jian Shi Technology (9877.HK), Health Yuan (JCARE.SW), Lepu Medical (LEPU.SW), Dingdang Health (9886.HK), Bio-Thera (2315.HK), Zhiyun Health (9955.HK), MeinGene (6667.HK), Prenetics (PRE.NASDAQ), Yunkang Group (2325.HK), Rike Biotech (2179.HK), Lepu Biotech (2157.HK), Clear Medical (1406.HK), Baxin An (2185.HK), Yonghe Medical (2279.HK), Kailaiying (6821.HK), Beihai Kangcheng (1228.HK), Gusheng Tang (2273.HK), Yingpeng Technology (2251.HK), Clover Biotech (2197.HK), Minimally Invasive Robotics (2252.HK), Harmony Cayman (2256.HK), Kunbo Medical (2216.HK), Xianruida (6669.HK), Kangsheng Global (9960.HK), Yimai Tong (2192.HK), Tengsheng BOP (2137.HK), Canopy (2162.HK), Chaoyu Eye Hospital (2219.HK), Guichuang Tongqiao (2190.HK), Huihuang Medicine (0013.HK), Kexi Pharmaceutical (2171.HK), Zhaoke Ophthalmology (6622.HK), Nature Pharmacy (UPC.NASDAQ), Sainfo Pharmaceutical (6600.HK), Zhaoyan New Drugs (6127.HK), Novogene Health (6606.HK), ADAG.NASDAQ, Beikang Medical (2170.HK), Jianbimiao Miao Miao (2161.HK), Minimally Invasive Xin Tong (2160.HK), Rui Li Medical Beauty (2135.HK), Jiaosisi Pharmaceutical (1167.HK), Hepbrium (2142.HK), JD Health (6618.HK), Deqi Pharmaceutical (6996.HK), Rongchang Biotech (9995.HK), WuXi AppTec (2126.HK), XianSheng Pharmaceutical (2096.HK), Yunding New Energy (1952.HK), Jiahe Biotech (6998.HK), Zai Ding Pharmaceutical (9688.HK), Ocular Biotech (1477.HK), Yongtai Biotech (6978.HK), Hapu Pharmaceutical (9989.HK), Kaopeng Pharmaceutical (9939.HK), Peijia Medical (9996.HK), Kangfang Biotech (9926.HK), Nuo Cheng Jianhua (9969.HK), Tianjing Biotech (IMAB.NASDAQ), Kanglong Chemical (3759.HK), China Antibody (3681.HK), Dongyao Pharmaceutical (1875.HK), Yasheng Medicine (6855.HK), Fuhong Hanlin (2696.HK), Hansoh Pharmaceutical (3692.HK), Mabtech (2181.HK), Fangda Holdings (1521.HK), Via Biotech (1873.HK), CStone Pharmaceuticals (2616.HK), Junshi Biosciences (1877.HK), WuXi AppTec (2359.HK), Innovent Biologics (1801.HK), Hailun Medicine (2552.HK), BeiGene (6160.HK), Galle Therapeutics (1672.HK), WuXi AppTec (2269.HK), China Resources Pharmaceutical (3320.HK), Yajie Scientific Research Pharmaceutical (2633.HK), Huihuang China Medicine (HCM.NASDAQ), GSK Biologics (1548.HK), BBI Life Sciences (1035.HK), etc. In terms of the number of filings, the Frost & Sullivan healthcare team maintains an absolute leading position in Hong Kong healthcare IPOs, consistently ranking first in market share from 2018 to 2022.
Since the listing of the first batch of companies on the Sci-tech Innovation Board in July 2019, Frost & Sullivan reports have been widely cited in the prospectuses of leading Sci-tech Innovation Board listed companies in the industry, including: Kangwei Century (688426.SH), Jinnan Protein (688137.SH), Nuo Cheng Jianhua (688428.SH), Aopu Mai Biotech (688293.SH), MicroPort Electrophysiology (688351.SH), Mengke Pharmaceutical (688373.SH), Yifang Biotech (688382.SH), Jicui Yaokang (688046.SH), Haichuang Pharmaceutical (688302.SH), Rongchang Biotech (688331.SH), Rendu Biotech (688193.SH), Shouyao Holdings (688197.SH), Heyuan Biotech (688238.SH), Yaxin Security (688225.SH), Xidi Micro (688173.SH), Mawei Biotech (688062.SH), Yahong Medicine (688176.SH), BeiGene (688235.SH), Jiahe Meikang (688246.SH), Dizhe Medicine (688192.SH), Novogene (688105.SH), Chengda Biotech (688739.SH), Geke Micro (688728.SH), Huaxi Biotech (688363.SH), Junshi Biotech (688180.SH), Zhejiang Oncology (688266.SH), BeiGene (688177.SH), Shenzhou Cells (688520.SH), etc. They are considered one of the most powerful, professional, and influential industry research institutions in the sector. We hope to work with enterprises to understand industry trends, seize development opportunities, jointly promote innovation and upgrading of China's healthcare industry, and build a healthy future.
Recommended Reading
Frost & Sullivan assists Lüzhoubio in successfully listing on the Hong Kong Stock Exchange (2480.HK)
Frost & Sullivan assists MesHealth in successfully listing on the Hong Kong Stock Exchange (2415.HK)
Frost & Sullivan assisted Yisheng Biology in successfully going public on the NASDAQ (NASDAQ:YS)
Frost & Sullivan assists Meiliyuan in successfully listing on the Hong Kong Stock Exchange (2373.HK)
Frost & Sullivan assisted IdeaChip to successfully go public in Hong Kong (1244.HK)
Frost & Sullivan helps Lepuventec successfully go public in Hong Kong (2291.HK)
Frost & Sullivan assists GenSense in successfully listing on the Hong Kong Stock Exchange (9877.HK)
Frost & Sullivan helps Bio-TheraPlex successfully list on the Hong Kong Stock Exchange (2315.HK)
Frost & Sullivan helps Zhiyun Health successfully go public in Hong Kong (9955.HK)
Frost & Sullivan assists MeinGene in successfully listing on the Hong Kong Stock Exchange (6667.HK)
Frost & Sullivan assisted Prenetics in successfully going public on the NASDAQ (NASDAQ:PRE)
Frost & Sullivan assists Clear Medical in successfully going public in Hong Kong (1406.HK)
Frost & Sullivan assists Baisin'an in successfully listing on the Hong Kong Stock Exchange (2185.HK)
Frost & Sullivan assists Kaleido in successfully listing on the Hong Kong Stock Exchange (6821.HK)
Frost & Sullivan helps Gushengtang successfully go public in Hong Kong (2273.HK)
Frost & Sullivan assisted Yuhuan in successfully listing on the Hong Kong Stock Exchange (2256.HK)
Frost & Sullivan assists Kunbo Medical in successfully going public in Hong Kong (2216.HK)
Frost & Sullivan assists ConocoAL in successfully listing on the Hong Kong Stock Exchange (2162.HK)
Frost & Sullivan helps Guichuangtongqiao successfully go public in Hong Kong (2190.HK)
Frost & Sullivan assists Nature Pharma in successfully going public in the US (NASDAQ):UPC)
Frost & Sullivan helps Jianbaimiao Miao successfully go public in Hong Kong (2161.HK)
Frost & Sullivan helps JD Health successfully go public in Hong Kong (6618.HK)
Frost & Sullivan assists Oculent in successfully listing on the Hong Kong Stock Exchange (1477.HK)
Frost & Sullivan helps develop PharmaCure successfully go public in Hong Kong (9939.HK)
Frost & Sullivan assisted Tianjing BIO in successfully going public on the NASDAQ (NASDAQ: IMAB)
Frost & Sullivan assisted ViaBio in successfully listing on the Hong Kong Stock Exchange (1873.HK)
Frost & Sullivan helps Cornerstone Pharmaceuticals successfully go public in Hong Kong (2616.HK)
Frost & Sullivan helps WuXi AppTec successfully go public in Hong Kong (2359.HK)
Frost & Sullivan helps BeiGene successfully go public in Hong Kong (6160.HK)
Frost & Sullivan helps WuXi AppTec Biologics successfully go public in Hong Kong (2269.HK)
Frost & Sullivan assisted Hua Huang China Medicine in successfully going public on NASDAQ (HCM)
*The above order is not sequential and is arranged in reverse chronological order based on listing time.

