
Haichuang Pharmaceutical Co., Ltd. (hereinafter referred to as 'Haichuang Pharmaceutical') successfully went public on April 12, 2022, issuing a total of 24.76 million shares at an issue price of 42.92 yuan per share.
Haichuang Pharmaceutical is an international innovative pharmaceutical company based on deuterated technology and PROTAC-directed protein degradation technology platforms. Its goal is to develop Best-in-class and First-in-class drugs with significant clinical needs. The company focuses on the research and development of innovative drugs for major therapeutic areas such as oncology and metabolic diseases. Adhering to the strategic concept of 'creating good medicines to benefit the world', it prioritizes providing patients with safe, effective, and affordable drugs, and is committed to developing and producing innovative drugs with global rights and interests.
The company emphasizes R&D innovation and has a research and development team with experience in the successful development of multiple new drugs, as well as work at well-known domestic and international pharmaceutical companies. The core technology team has in-depth research and understanding of the mechanisms of major diseases such as cancer and metabolic disorders, and possesses rich experience and scientific sensitivity in target selection, compound design, biomodel design, preclinical research, clinical protocol design, and clinical studies. Since its establishment, the company has persisted in independent innovation. Through R&D accumulation, it has established a deuterated drug R&D platform, a PROTAC targeted protein degradation technology platform, a targeted drug discovery and validation platform, and a lead compound optimization screening platform. It has built a mature technical system from early discovery to clinical development of innovative drugs, which can improve the success rate of the company's ongoing research products and shorten the drug R&D cycle in aspects such as target discovery and validation, lead compound discovery, drug molecule design, chemical synthesis process and formulation development, clinical protocol design and development. The company's core products include the AR inhibitor deuterated enzalutamide, which has received support from the national 'Major New Drug Creation' science and technology special project, the HC-1119 product, as well as the URAT1 inhibitor and HP501 product independently developed by the company for the treatment of hyperuricemia and gout. Frost & Sullivan has long been paying attention to the global and Chinese biopharmaceutical industries, publishing a large number of research reports, which are widely cited in the prospectuses of leading companies listed on the Sci-Tech Innovation Board, helping customers accelerate growth.
Global and China Anti-Tumor Drug Market Overview
The global anti-tumor drug market is booming, with a market size reaching $150.3 billion in 2020. It is expected that by 2025, the market size will reach $304.8 billion, with a compound annual growth rate of 15.2% from 2020 to 2025. By 2030, the anti-tumor drug market will further grow to $482.5 billion, with a compound annual growth rate of 9.6% from 2025 to 2030.
Global market size of anti-tumor drugs2016-2030E
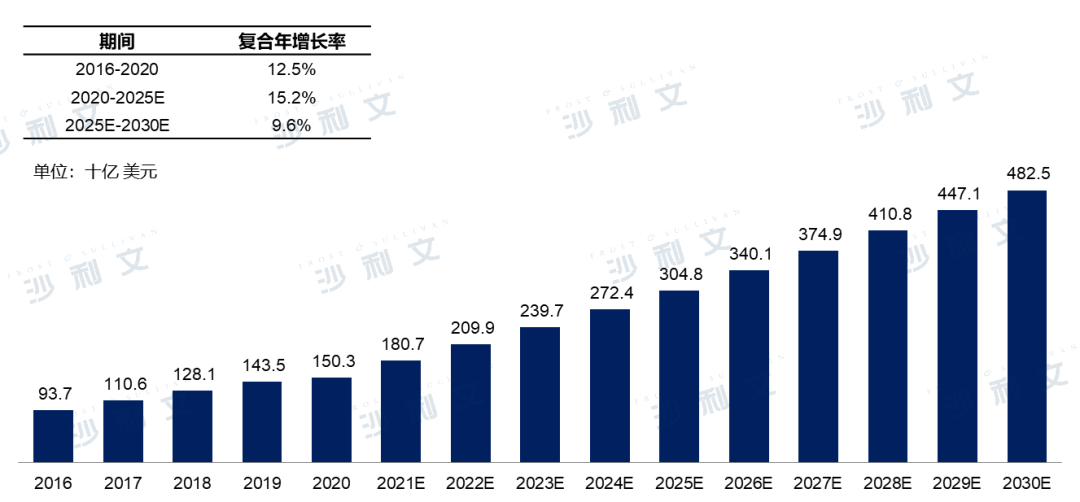
Source: Frost & Sullivan report
In the Chinese pharmaceutical market, the sales of anti-tumor drugs have been showing a steady growth trend in recent years. In 2020, the market size reached RMB 1975 billion. The progress in cancer treatment methods has prompted the Chinese anti-tumor drug market to continue on an upward trajectory in the coming years. It is expected that the Chinese anti-tumor drug market will reach RMB 4162 billion by 2025, with a compound annual growth rate of 16.1% from 2020 to 2025, and reach RMB 6831 billion by 2030.
Market scale of anti-tumor drugs in China2016 - 2030E
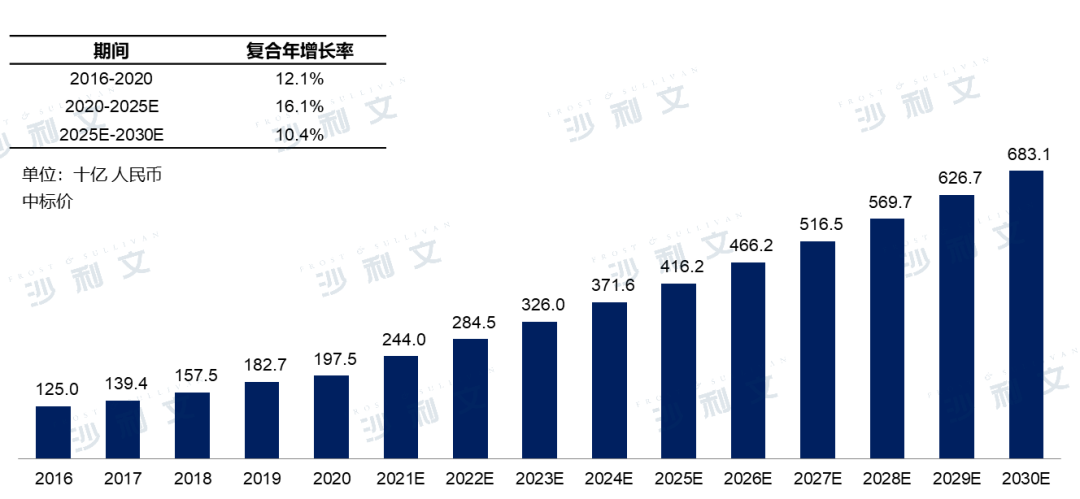
Source: Frost & Sullivan report
Global and China Prostate Cancer Drug Market Overview
Currently, the global prostate cancer drug market size has grown from $8.9 billion in 2016 to $13.9 billion in 2020, with an annual compound growth rate of 11.7%. It is expected that by 2025, the market size will reach $26.6 billion, with an annual compound growth rate of 13.8%. By 2030, the global prostate cancer drug market will further grow to $37.6 billion, with an annual compound growth rate from 2025 to 2030 of 7.2%.
Global prostate cancer market size and forecast, 2016-2030E
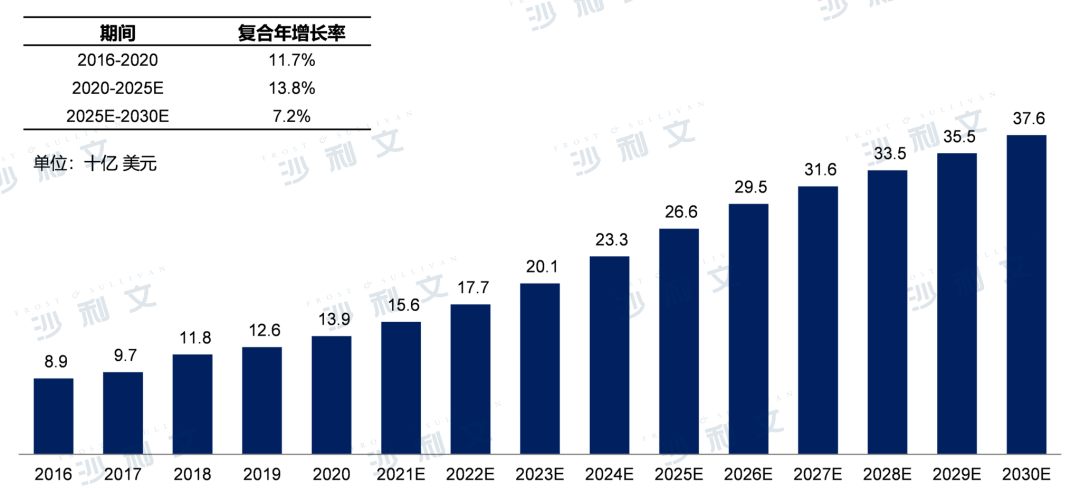
Source: Frost & Sullivan report
Currently, the market scale of prostate cancer drugs in China has grown from 2.6 billion yuan in 2016 to 5.6 billion yuan in 2020, with an annual compound growth rate of 21.0%. It is expected that by 2025, the market scale will reach 212 billion yuan, with an annual compound growth rate of 30.7%. By 2030, the prostate cancer drug market in China will further grow to 506 billion yuan, with an annual compound growth rate of 19.0% from 2025 to 2030.
Market scale and forecast of prostate cancer in China, 2016-2030E
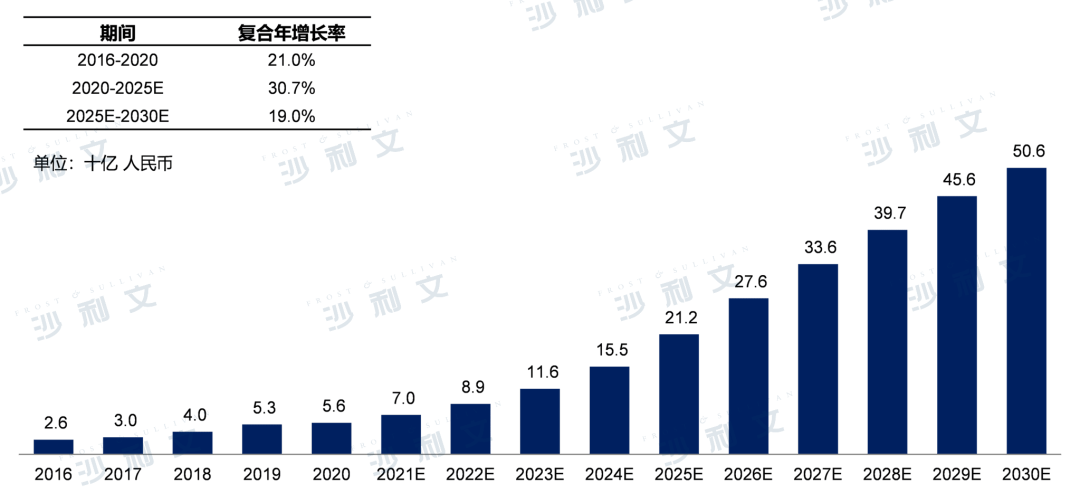
Source: Frost & Sullivan report
Global and China Metabolic Disease Drug Market Overview
In the global metabolic disease drug market, the size of the market has been steadily growing in recent years. From 2016 to 2020, the market size reached $105.4 billion in 2020, with a compound annual growth rate of 3.3% over the past five years. It is estimated that the global metabolic disease drug market will reach $149 billion in 2025, with an annual compound growth rate of 7.2%, and reach $2121 billion by 2030.
Global market size of metabolic disease drugs, 2016 - 2030E
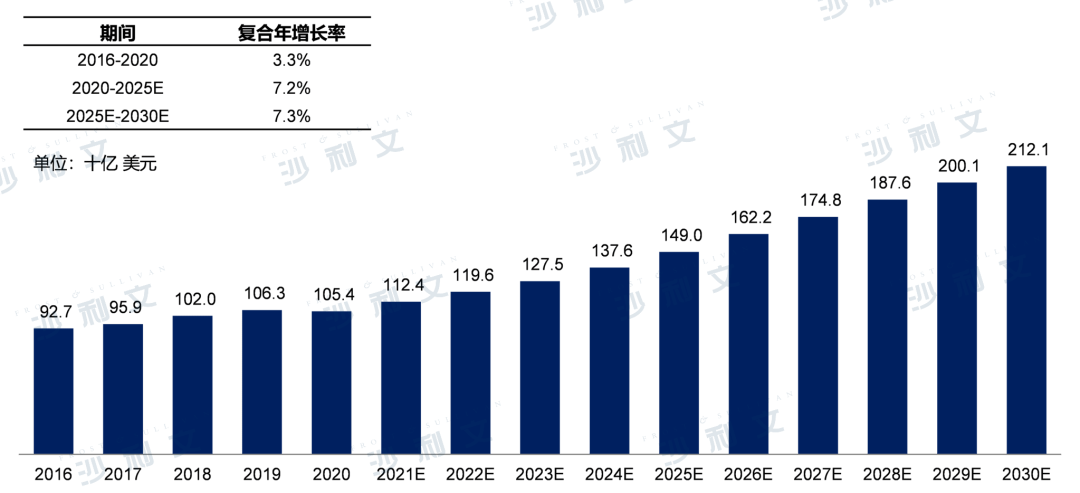
Source: Frost & Sullivan report
From 2016 to 2020, the market size of metabolic disease drugs in China reached RMB 922 billion in 2020, with a compound annual growth rate of 8.4% over the past five years. It is estimated that the market for metabolic disease drugs in China will reach RMB 1654 billion by 2025, with an annual compound growth rate of 12.4%, and reach RMB 2829 billion by 2030.
Market Size of Metabolic Disease Drugs in China, 2016-2030E
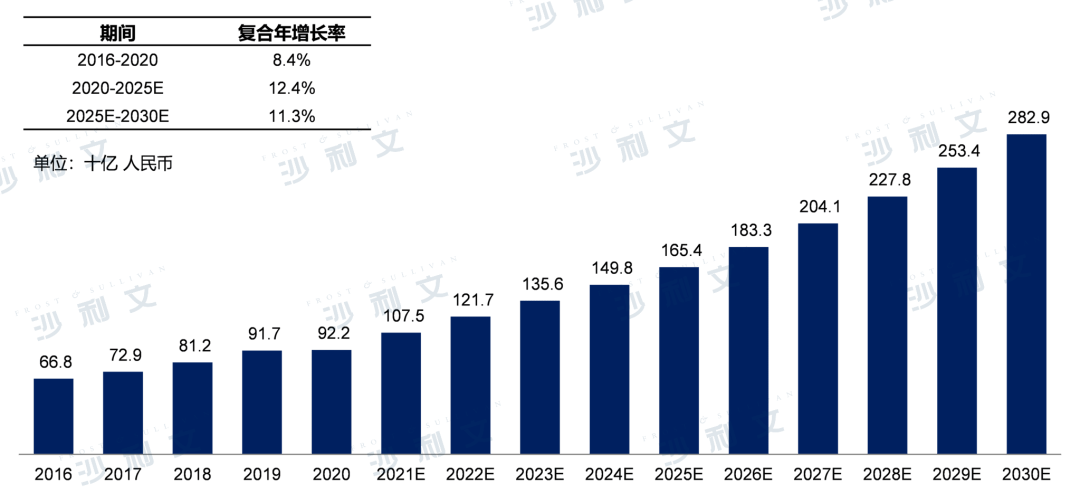
Source: Frost & Sullivan report
Global and China Hyperuricemia/Gout Market Overview
Currently, the global market size for gout medications has decreased from $28 billion in 2016 to $25 billion in 2020, with a compound annual growth rate of -2.7%. It is expected that the global market for hyperuricemia/gout will continue to grow from 2020 onwards, reaching a market size of $37 billion by 2025, with a compound annual growth rate of 7.9%. By 2030, the global gout medication market will further grow to $77 billion, with a compound annual growth rate of 16.0% from 2025 to 2030.
Global market size of gout medications, 2016 - 2030E
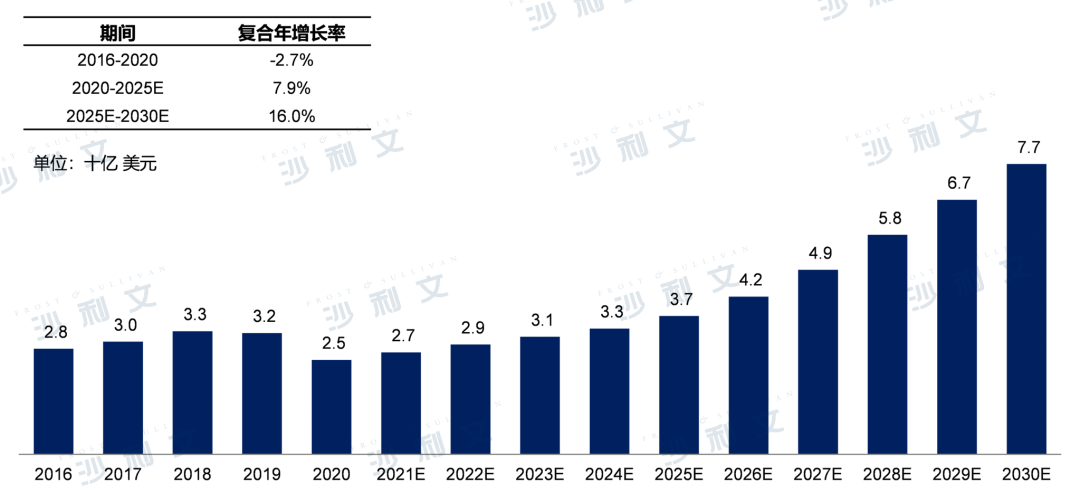
Source: Frost & Sullivan report
Currently, the market scale of gout medications in China has grown from RMB 900 million in 2016 to RMB 2.7 billion in 2020, with an annual compound growth rate of 29.9%. It is expected that by 2025, the market scale will reach RMB 29 billion, with an annual compound growth rate of 1.3%. By 2030, the gout medication market in China will further grow to RMB 108 billion, with an annual compound growth rate of 30.5% from 2025 to 2030.
Gout medication market size in China, 2016-2030E
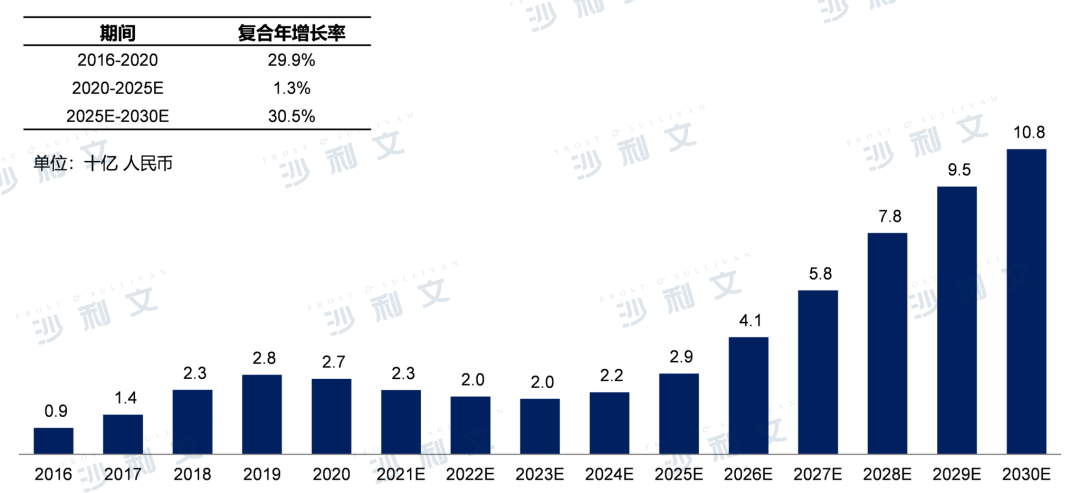
Source: Frost & Sullivan report
Deuterium technology overview
Deuterated drugs are a class of medications that replace hydrogen atoms at specific sites in the molecular structure of marketed original drugs with deuterium atoms. The main purpose of such drug modification is to reduce the frequency of administration, improve patient medication compliance, achieve the same therapeutic effect with a smaller dose, reduce adverse drug reactions, and clinically benefit patients without sacrificing the activity of the original drug.
Deuterated compounds are new drugs that can obtain patent authorization. The advantages of deuterated drugs can be subdivided into the following points: 1) Increase drug exposure and prolong the duration of action in the body. 2) Reduce specific metabolites and improve metabolic profiles: The antidepressant paroxetine can form highly active metabolites in the body. 3) Reduce mutual conversion and stabilize stereoisomers. 4) Reduce first-pass effects and increase oral bioavailability.
PROTAC Technology Overview
Proteolysis targeting chimera (PROTAC) is a new technology based on the ubiquitin-proteasome system that induces the degradation of target proteins through small molecule compounds. Traditional small molecule drugs and monoclonal antibodies need to continuously occupy the active site of the target protein to block its function, which is an 'occupancy driven' approach. Such drugs require certain dosage, a long enough half-life, high affinity, and other conditions, making them prone to side effects, off-target toxicity, and resistance. PROTAC, on the other hand, only provides binding activity, triggering the event of binding between the target protein and E3 ligase to initiate degradation, which is an 'event driven' approach. It does not directly inhibit the functional activity of the target protein, nor does it require long-term and high-intensity binding with the target protein. Therefore, it can target proteins with smooth surfaces lacking small molecule binding regions, as well as many targets that cannot be regulated by small molecules or reached by antibodies.
Frost & Sullivan, integrating 61 years of global consulting experience, has dedicated 24 years to serving the booming Chinese market. With a global perspective, we help clients accelerate their business growth and achieve benchmark positions in industry growth, innovation, and leadership. The healthcare industry is one of Frost & Sullivan's core areas of focus. Over the past seventeen years, our healthcare team has provided financing financial advisory, IPO industry advisory, strategic consulting, management consulting, and other services to hundreds of outstanding domestic and international biopharmaceutical, medical device, healthcare service, and internet healthcare companies. Successful listings include: Ruike Biotech (2179.HK), Leopu Biotech (2157.HK), Clear Medical (1406.HK), Bexian An (2185.HK), Yonghe Medical (2279.HK), Kailaiying (6821.HK), Beihai Kangcheng (1228.HK), Gusheng Tang (2273.HK), Yingtong Technology (2251.HK), Clover Biotech (2197.HK), Minimally Invasive Robotics (2252.HK), Huiyuan Kamman (2256.HK), Kunbo Medical (2216.HK), Xianruida (6669.HK), Kangsheng Global (9960.HK), Yimaitong (2192.HK), Tengsheng Bopharm (2137.HK), Canopy (2162.HK), Chaoyu Ophthalmology (2219.HK), Guichuang Tongqiao (2190.HK), Hua Huang Medicine (0013.HK), Koi Pharmaceutical (2171.HK), Zhaoke Ophthalmology (6622.HK), Nature Pharmaceuticals (UPC.NASDAQ), Saiseng Pharmaceutical (6600.HK), Zhaoyan New Drugs (6127.HK), Novogene Health (6606.HK), ADAG.NASDAQ, Beikang Medical (2170.HK), Jianbimiao Miao (2161.HK), Minimally Invasive Xin Tong (2160.HK), Rui Li Medical Beauty (2135.HK), Jiake Pharmaceutical (1167.HK), Hepcon Pharma (2142.HK), JD Health (6618.HK), Deqi Pharma (6996.HK), Rongchang Biotech (9995.HK), WuXi AppTec (2126.HK), Siasun Pharmaceutical (2096.HK), Yunding New Energy (1952.HK), Jiahe Biotech (6998.HK), Zai Ding Pharma (9688.HK), Ocular Care Biologics (1477.HK), Yongtai Biotech (6978.HK), Hapu Pharma (9989.HK), Kuaibo Pharmaceutical (9939.HK), Peijia Medical (9996.HK), Kangfang Biotech (9926.HK), Nuocheng Jianhua (9969.HK), IMAB.NASDAQ, Kanglong Chemical (3759.HK), China Antibody (3681.HK), Dongyao Pharmaceutical (1875.HK), Yasheng Pharmaceutical (6855.HK), Fuhong Hanlin (2696.HK), Hansoh Pharmaceutical (3692.HK), Mabtech Pharma (2181.HK), Fangda Holdings (1521.HK), Via Bio (1873.HK), CStone Pharmaceuticals (2616.HK), Junshi Biosciences (1877.HK), WuXi AppTec (2359.HK), Innovent Biologics (1801.HK), Hualing Medicine (2552.HK), BeiGene (6160.HK), Gilead Sciences (1672.HK), WuXi AppTec (2269.HK), China Resources Pharmaceutical (3320.HK), Jacobus Buchen Research & Pharma (2633.HK), Hua Huang China Medicine (HCM.NASDAQ), Biotechnology (1548.HK), BBI Life Sciences (1035.HK), and others. In terms of the number of listed projects, our healthcare team maintains an absolute leading position in Hong Kong's healthcare IPO market, consistently ranking first in market share from 2018 to 2021.
Since the listing of the first batch of companies on the Sci-tech Innovation Board in July 2019, Frost & Sullivan reports have been widely cited in the prospectuses of leading Sci-tech Innovation Board listed companies in the industry. This includes Rongchang Biotech (688331.SH), Rendu Biotech (688193.SH), Shouyao Holdings (688197.SH), Heyuan Biotech (688238.SH), Yaxin Security (688225.SH), Xidi Micro (688173.SH), Mawei Biotech (688062.SH), Yahong Medicine (688176.SH), BeiGene (688235.SH), Jiahe Meikang (688246.SH), Dizhe Medicine (688192.SH), Novogene (688105.SH), Chengda Biotech (688739.SH), Geke Micro (688728.SH), Huaxi Biotech (688363.SH), Junshi Biosciences (688180.SH), Zhejiang Genomics & Therapeutics (688266.SH), BeiGene (688177.SH), and Shenzhou Cells (688520.SH). They are considered to be the most powerful, professional, and influential industry research institutions in the sector. We hope to work with enterprises to understand industry trends, seize development opportunities, jointly promote innovation and upgrading of China's healthcare industry, and build a healthy future.
Recommended Reading
23. Frost & Sullivan assists Nature Pharma in successfully going public in the US (UPC.NASDAQ)
Frost & Sullivan helps Zhaoyan New Drugs successfully list on the Hong Kong Stock Exchange (6127.HK)
41. Frost & Sullivan assists Zai Lab to successfully go public in Hong Kong (9688.HK)
49. Frost & Sullivan assisted Tianjing Biology in successfully going public in the US (IMAB.NASDAQ)

